Light exposure management is the next thing we need to discuss in this series on foundational sleep hygiene. If you have been following along, we’ve already discussed the importance of tracking your sleep, we’ve also established that consistent sleep and wake times give your circadian rhythm predictability, and that a proper wind down routine transitions your nervous system from activation to rest. But there’s another element that’s arguably more powerful than everything we have discussed so far, and it’s also (surprisingly) the one most people completely overlook: light exposure management.
Light is the primary zeitgeber (time-giver) for your circadian rhythm. It’s the signal your brain uses to determine what time it is, when to be alert, and when to sleep. Everything else we’ve discussed works better when your light exposure is optimised. And almost everything works worse when it’s not.
Frustratingly, modern life has completely divorced us from the natural light-dark cycle that our biology evolved expecting. We spend our days indoors under artificial light that’s far dimmer than outdoor daylight. We spend our evenings and nights under artificial light that’s far brighter than our ancestors ever experienced after sunset. We stare at screens that emit the exact wavelength of light our brains interpret as “morning sun, stay alert.” And then we wonder why we can’t fall asleep when we want to and why we feel groggy when we wake.
The solution isn’t complicated in principle, you simply try to get lots of bright light during the day, especially morning, and very little light in the evening, especially the blue wavelengths. But implementation collides with how we actually live: working indoors all day, using screens for both work and entertainment, sharing space with people who have different schedules, living in climates where winter means limited daylight, etc.
So, the aim of this article is to give you a comprehensive understanding of light exposure management; why it matters at a level deeper than “blue light is bad,” what actually constitutes effective light exposure (because the details matter enormously), and how to implement this in real life with all its constraints and complications. This is where theory meets the practical reality of trying to optimise biology while living in the modern world.
Table of Contents
- 1 Why Morning Light is the Single Most Important Thing For Sleep
- 2 Getting Morning Light: Practical Implementation
- 3 Daytime Light
- 4 Evening Light Reduction
- 5 Blue Light: Why Wavelength Matters As Much As Intensity
- 6 Practical Evening Light Strategies: Light Exposure Management Implementation
- 7 Screen Management
- 8 Blue Light Blocking Glasses
- 9 Special Considerations: The Details That Matter
- 10 Light Exposure and Shift Work: Strategic Use
- 11 Measuring Light
- 12 Troubleshooting Common Light Exposure Management Obstacles
- 13 Light Exposure Management Conclusion
- 14 Author
Why Morning Light is the Single Most Important Thing For Sleep
If you could only implement one intervention from this entire sleep series, and if you had to choose a single practice that would have the greatest impact on your sleep quality, morning light exposure would be a very strong contender for it. Not your bedtime routine, not your wake time, not reducing evening screens. Morning light.
The reason is that morning light is the primary signal your circadian rhythm uses to calibrate itself each day. Your circadian rhythm has a natural period that’s slightly longer than twenty-four hours for most people (typically around twenty-four hours and fifteen minutes). Without regular calibration, it would drift later and later. Morning light is how your body synchronises this internal clock to the actual twenty-four-hour day.
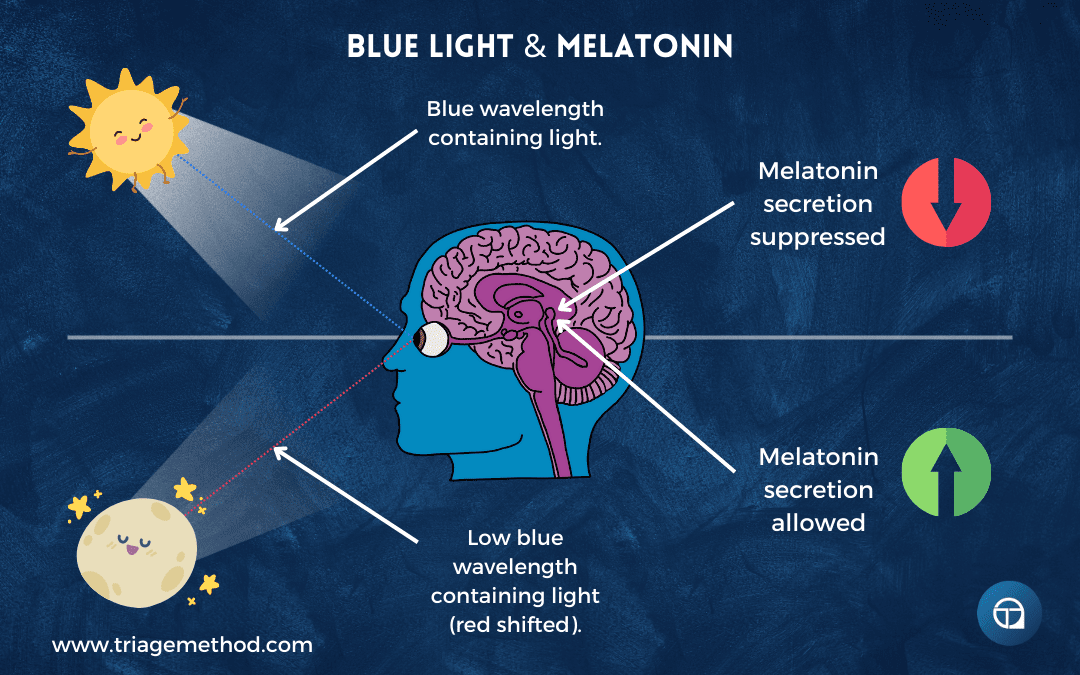
When you expose your eyes to bright light in the morning (and we’ll get very specific about “bright” shortly), you’re activating specialised photoreceptors in your retina called intrinsically photosensitive retinal ganglion cells (ipRGCs). These cells contain a photopigment called melanopsin that’s most sensitive to blue light around 480 nanometers, which is the wavelength present in morning and midday sunlight.
These cells send signals directly to your suprachiasmatic nucleus (SCN), the master circadian clock in your brain. This signal essentially says “It’s morning. The day has begun. Set the clock accordingly.” In response, your SCN coordinates a cascade of changes throughout your body: cortisol rises appropriately, body temperature begins climbing from its nighttime low, alertness increases, and crucially, it sets a timer for when melatonin should begin rising again roughly fourteen to sixteen hours later.
This last point is super important. Morning light doesn’t just help you wake up. It determines when you’ll get sleepy that evening. If you get strong morning light at 7am, your body will begin preparing for sleep around 9-10pm. If you don’t get morning light until 10am, your sleep preparation won’t begin until midnight or 1am. If you get weak, inconsistent light throughout the morning, your circadian rhythm remains uncertain about what time it actually is, and you might not get a clear sleep signal at all.
The timing matters enormously. You want this light exposure within thirty to sixty minutes of waking. During this window, your circadian system is most responsive to the calibrating effect of light. Light exposure at 7am has a much stronger effect than the same light at 10am. If you wake at 7am but don’t get bright light until 9am, you’ve missed the optimal window, and your circadian rhythm remains somewhat ambiguous about when your day actually began.
How much light is enough? This is where most people drastically underestimate what’s required. Typical indoor lighting like your kitchen lights, office lights, and even bright rooms, provides 300-500 lux. This is far too dim to provide a meaningful circadian signal. Your circadian system evolved in an environment where daytime meant tens of thousands of lux from the sun.
For effective circadian entrainment, you need at minimum 1,000 lux, ideally 10,000+ lux. To put this in perspective:
- Typical indoor lighting: 300-500 lux
- Bright office space: 500-1,000 lux
- Overcast day outdoors: 10,000 lux
- Direct sunlight (not looking at sun): 50,000-100,000 lux
The difference between indoor and outdoor light is gigantic. Even on a heavily overcast winter day in London (where I am writing from this morning), you’re getting ten times more light outdoors than in a bright indoor space. On a clear day, you’re getting fifty to one hundred times more light.
This means that standing near a window, while better than nothing, is not sufficient if you’re serious about optimising your circadian rhythm. The light intensity drops off dramatically as you move away from the window, and glass filters some of the beneficial wavelengths. You need to actually go outside, even if only briefly.
Duration: Ten to thirty minutes of bright light exposure is the target, depending on light intensity. If you’re getting direct sunlight on a clear day (50,000+ lux), ten minutes is sufficient. If it’s overcast (10,000 lux), aim for twenty to thirty minutes. If you’re using indoor light therapy boxes (usually 10,000 lux), thirty minutes is standard.
You don’t need to stare at the light or the sky, and in fact, you should probably never look directly at the sun, as it can damage your eyes. The light just needs to reach your eyes. You can be having breakfast, checking your phone, going for a walk, sitting and reading. Whatever you’re doing, just do it in bright light.
Even on cloudy days. I know it’s tempting to think, “It’s grey and miserable, there’s no point going outside.” But remember that overcast outdoor light is still 10,000 lux, which is twenty times brighter than your indoor spaces. Your circadian system doesn’t care that it feels dark to you. The light meters in your retinas are getting a clear signal that it’s daytime.
Even if you don’t “feel” it working. You won’t feel more alert immediately from morning light the way you do from coffee. The effect is subtle in the moment but profound over time. You’re setting your circadian clock, not getting an acute stimulant effect. Trust the process even when you can’t feel anything happening.
This is perhaps the highest-leverage intervention available because it’s addressing the root mechanism (your circadian rhythm) rather than just treating symptoms. Get morning light right, and falling asleep becomes easier, wake time becomes less brutal, daytime alertness improves, and the entire system starts working with you instead of against you.
Getting Morning Light: Practical Implementation
Now, the gold standard for light exposure here is simple: go outside within thirty minutes of waking and stay outside for ten to thirty minutes. Take your coffee outside. Eat breakfast on your patio or balcony. Go for an early morning walk. Stand in your garden while you check your phone. The activity doesn’t matter. The light does.
If you exercise in the morning, do it outside or at least try to commute there with outside light in mind. A morning run, walk, or even just standing outside while you do some mobility work gives you the double benefit of movement (which promotes better sleep) and light exposure (which entrains your circadian rhythm). This is one of the most powerful habit stacks available: wake at the same time every day, immediately get bright light, and incorporate movement. If you can establish this pattern, you’ve solved most sleep problems before they start. I know that isn’t what most people want to hear, but it is the truth.
Now, if going outside isn’t immediately practical (e.g. you need to prepare breakfast for children, you have a long morning routine, you live somewhere genuinely unsafe or inhospitable in early morning), then prioritise getting outside within the first hour instead of the first thirty minutes. Every minute of delay reduces the effectiveness somewhat, but light at 7:45am is still far better than light at 10am or no meaningful light exposure at all.
When you can’t get outside early, bring the outdoors in as much as possible. Open curtains and blinds immediately upon waking. If you have a window near where you eat breakfast or get ready, position yourself there. The closer you are to a window, the more light reaches your eyes. Sitting within a meter of a window on a bright day can provide several thousand lux. This is not as good as being outside, but significantly better than internal rooms.
If you work from home, consider setting up your morning workspace near a window. Those first few hours of your workday can provide the light exposure you need if outdoor time isn’t feasible. This won’t work if your window faces north or is shaded by buildings, but a south-facing window (in the Northern Hemisphere) or any unshaded window can provide substantial light.
Light therapy boxes are the final option when natural light genuinely isn’t available. There are multiple situations where these may actually even be your best option: if you wake before sunrise in winter, if you work night shifts and need to shift your circadian rhythm, or if you live in high latitudes with extended darkness. These devices provide 10,000 lux at a specific distance (usually 30-40 centimetres) and are clinically validated for treating seasonal affective disorder.
To use a light therapy box effectively, you need to position it at roughly eye level, 30-40 centimetres away, and slightly to the side rather than directly in front, so you’re not staring into it. The light needs to reach your eyes, but you don’t need to look at it continuously. Most people use them while eating breakfast, working, or reading. Thirty minutes daily is the standard protocol.
Light therapy boxes are relatively cheap, especially for something you will use daily and for years. If you genuinely cannot get outdoor morning light regularly, particularly in winter, this is money well spent. But for most people, they should be considered a backup option, not a replacement for natural daylight. A grey winter morning outdoors is still better than artificial light indoors.
Consistency is absolutely critical. Getting morning light occasionally doesn’t entrain your circadian rhythm. Your body needs the signal every day, seven days a week, including weekends. This is where many people fall down. They’re disciplined about morning light Monday through Friday but skip it on weekends, which undermines the entire pattern. Your circadian rhythm doesn’t distinguish between weekdays and weekends. It needs consistent calibration.
This is another place where consistent wake times and morning light exposure reinforce each other. When you wake at the same time every day and immediately get light exposure, you’re providing your circadian system with the strongest and most reliable signal possible. Your body learns exactly when the day begins, and everything else follows from that foundation.
Daytime Light
Morning light gets the attention because it’s most important for circadian entrainment, but maintaining bright light exposure throughout the day matters too. This is where modern indoor living creates enormous problems.
When you spend your entire day in 300-500 lux environments, your circadian system loses the distinction between day and night. If daytime light levels are only three times brighter than your evening light, that’s not sufficient contrast. Your brain needs to experience a dramatic difference between day (very bright) and night (very dim) to maintain clear circadian rhythms.
People who work indoors all day with minimal outdoor exposure often find they’re not particularly sleepy at bedtime, even with good wind-down routines and reduced evening light. The reason is that their daytime light exposure was too similar to their evening light exposure. They haven’t created enough contrast.
The solution isn’t to make your indoor workspace blindingly bright, as that’s not practical and would be uncomfortable. The solution is to get outdoor breaks during the day. Lunch outside, even ten to fifteen minutes, provides a substantial light boost. A short walk mid-morning or mid-afternoon. Even standing outside for five minutes between meetings. These bursts of high-intensity light help maintain the day-night distinction.
If outdoor breaks genuinely aren’t possible, and sometimes they’re genuinely not, you should try to position your workspace near windows. South-facing windows (Northern Hemisphere) provide the most consistent bright light throughout the day. Consider whether you can arrange your workspace to maximise natural light exposure. This might not be under your control in traditional offices, but if you’re working from home or have any influence over workspace design, prioritise natural light.
Avoiding dim spaces during the day is equally important as seeking bright spaces. Spending hours in windowless meeting rooms, dark offices, or consistently working in spaces with poor lighting suppresses your daytime alertness and reduces circadian contrast. When you have a choice, choose bright over dim during daytime hours.
Daytime light exposure affect nighttime sleep so much because your circadian rhythm isn’t just responding to the presence or absence of light, it’s actually integrating light information across the entire day to determine your phase. Strong daytime light makes your circadian system more robust and more resistant to the disrupting effects of evening light. It’s like giving your circadian clock a stronger signal to work with, which makes it less confused by weaker contradictory signals later.
People with strong daytime light exposure can often “get away with” more evening light exposure without significant circadian disruption. Their circadian system has such a clear daytime signal that moderate evening light doesn’t override it as easily. Conversely, people with weak daytime light exposure find their circadian rhythm is fragile and easily disrupted by even modest evening light.
This doesn’t mean you should use daytime light exposure as permission to ignore evening light management. You shouldn’t. But it does mean that if you’re struggling with sleep despite good evening light hygiene, insufficient daytime light might be the missing piece.
Evening Light Reduction
You’ve heard it a thousand times: avoid screens before bed, dim the lights, reduce blue light exposure. You know this. Everyone knows this. And yet, scroll through any social media platform at 10pm, and you’ll find millions of people bathing their retinas in blue light whilst wondering why they can’t sleep.
The gap between knowing and doing is vast, and part of why people fail to implement evening light reduction is that they don’t understand just how sensitive their circadian system is to light after dark. They think “I’ll just dim my phone and it’ll be fine” or “It’s just an hour of TV, it can’t matter that much.” But it does matter, far more than most people realise.
Your circadian system is asymmetric in its response to light. It takes a lot of bright morning light to shift your rhythm earlier, but relatively little evening light to shift it later. The mechanisms are different: morning light activates your circadian system through melanopsin in those ipRGCs we discussed previously. Evening light suppresses melatonin production directly. It’s much easier to suppress melatonin than to advance your circadian phase, which is why evening light has such a disproportionate impact.
Even relatively dim light (50 to 100 lux, which is a dimly lit room) can suppress melatonin production by 10-20%. Moderate light (200-300 lux, which is typical ambient lighting in homes) can suppress melatonin by 40-60%. And bright light (screens, overhead lights, anything above 500 lux) can suppress melatonin production by 80-90% or more.
This matters because melatonin isn’t just “the sleep hormone” that makes you feel sleepy. It’s the signal that communicates to your entire body that it’s nighttime. When melatonin is suppressed, you’re not just less sleepy; you’re telling every cell in your body that it’s still daytime, which affects everything from body temperature to immune function to cognitive performance.
The recommendation to start reducing light exposure two to three hours before bed isn’t arbitrary. It’s based on how long melatonin takes to rise after you stop suppressing it. If your target bedtime is 10:30pm and you’re exposed to bright light until 10pm, your melatonin hasn’t had time to rise when you try to sleep. You need the light reduction to begin by 7:30 or 8pm at the latest to allow melatonin to reach sleep-promoting levels by bedtime.
The ideal pattern is gradual dimming: normal indoor lighting until early evening (6-7pm), dimmer lighting from early evening through your wind-down period (7-9pm), and very dim lighting (ideally under 10 lux) in the final hour before bed (9:30-10:30pm if your bedtime is 10:30pm).
Ten lux is extraordinarily dim. It’s candlelight. It’s a small nightlight. It’s darker than most people normally experience in their homes in the evening. This is the level at which melatonin suppression becomes negligible. Anything brighter suppresses melatonin to some degree.
I’m not suggesting you literally need to live by candlelight in the final hour, although if you tried it, you’d probably sleep remarkably well. I’m suggesting that you should aim for very dim lighting in the final wind-down hour, understanding that every lux of additional light makes falling asleep somewhat harder.
Blue Light: Why Wavelength Matters As Much As Intensity
When people talk about light and sleep, they often focus exclusively on blue light. “Just avoid blue light and you’ll be fine.” This is both true and misleading. Blue light is the primary culprit, but understanding why helps you make better decisions about light exposure management.
Remember those melanopsin-containing cells in your retina that signal your circadian clock? They’re most sensitive to light around 480 nanometers, which is squarely in the blue part of the spectrum. Morning sunlight is rich in these wavelengths, which is why it’s so effective at entraining your rhythm. But so are most modern light sources: LED lights, fluorescent lights, and particularly screens.
The peak sensitivity is around 460-480 nanometers. Light at these wavelengths is roughly five times more effective at suppressing melatonin and activating your circadian system than light at other wavelengths of equivalent intensity. This means that 100 lux of blue light has similar circadian effects to 500 lux of red light.
This is why screens are particularly problematic. They emit substantial amounts of blue light directly toward your eyes from short distances. A phone screen at typical brightness can provide 200-300 lux to your retinas even if the ambient room lighting is much dimmer. You’re essentially shining a small daylight simulator directly at your circadian clock for hours every evening.
The blue-yellow colour balance of light matters as much or more than total intensity. Warm-colored light (red, orange, amber) has minimal blue wavelengths and therefore minimal circadian-disrupting effects. You can tolerate much brighter levels of warm light in the evening without significant melatonin suppression compared to cool white or blue-rich light.
This is why firelight, candlelight, and incandescent bulbs at lower temperatures (2200-2700K) are sleep-friendly: they emit predominantly in the red-orange-yellow spectrum with minimal blue. Modern LED lights, even “warm white” ones, often have a spike of blue wavelengths in their spectrum because that’s how white LEDs work; they’re actually blue LEDs with a phosphor coating that converts some blue light to other colours. The result is white light, but with more blue content than traditional incandescent bulbs.
This explains why your grandmother’s house with its old incandescent bulbs might have felt more conducive to evening relaxation than your modern home with its energy-efficient LEDs. The newer technology is better for energy efficiency and arguably better for daytime use, but worse for evening use because of the blue content.
Red and amber light are genuinely sleep-friendly because they contain almost no blue wavelengths. You can have relatively bright red or amber light in the evening (even 200-300 lux) with minimal circadian disruption. This is why some people use red night lights, amber bulbs, or even red LED strips for evening lighting. It provides enough light to see without suppressing melatonin.
The catch is that red light makes everything look weird. Colours are distorted, reading becomes more difficult, and you’re essentially living in a perpetual sunset. Some people find this atmosphere lovely and calming. Others find it strange and off-putting. There’s no avoiding the trade-off: you can have normal-looking light that disrupts sleep, or sleep-friendly light that looks abnormal.
Practical Evening Light Strategies: Light Exposure Management Implementation
Understanding the science is one thing. Actually modifying your evening light environment while living in the real world is another. Let’s get practical about what works for light exposure management.
Dimmer switches are a hugely valuable investment you can make for evening light management. Installing dimmers on your main evening living spaces (living room, bedroom, and bathroom) gives you control over light intensity rather than the binary choice of full brightness or darkness. You can gradually dim lights throughout your wind-down period, mimicking the natural transition from day to dusk to night.
Most dimmer switches are super cheap and are straightforward to install if you’re moderately handy. Smart bulbs (Philips Hue, LIFX, etc.) offer dimming capability along with colour temperature control, but they’re more expensive. The investment pays off immediately in improved sleep quality, particularly if you’re someone who struggles with sleep onset.
Using lamps instead of overhead lights instantly reduces light intensity because lamps provide localised lighting rather than flooding the entire room. Overhead lights are designed for visibility and activities like cleaning, cooking, and socialising. Evening wind-down doesn’t require that level of illumination. A single reading lamp or a few strategically placed table lamps provide adequate light for most evening activities while dramatically reducing total light exposure.
Choose warm-coloured bulbs (2200-2700K colour temperature) for your evening lamps. Look for labels saying “warm white,” “soft white,” or “amber.” Avoid “cool white,” “daylight,” or “bright white” bulbs in evening spaces as these have higher blue content. Specialist sleep-friendly bulbs are available that specifically filter blue wavelengths, though they’re more expensive and often cast a noticeably orange tint.
Salt lamps have become popular for evening lighting, and whilst the claims about “negative ions” and health benefits are largely unsupported, they do provide very dim, warm-colored light that’s genuinely sleep-friendly. A typical salt lamp provides 5-20 lux of orange-amber light, which is enough to navigate a room without stumbling but not enough to suppress melatonin significantly. They’re not sufficient as primary evening lighting if you’re trying to read or do activities, but they work well as ambient background lighting.
Candles provide the ultimate sleep-friendly light as they are warm spectrum, very low intensity, and have a flickering pattern that’s inherently calming. Our ancestors spent thousands of years experiencing fire as the only light source after dark, and we’re adapted to it. However, the obvious constraint is safety. Never leave candles unattended, keep them away from flammable materials, and extinguish them before sleep. For some people, the anxiety about fire safety outweighs the sleep benefits. For others, candlelit evenings become a cherished part of their wind-down ritual. They do also produce CO2, which many homes (especially modern homes) are not well designed to eliminate. So, if you are using candles, make sure you have a well-ventilated home.
String lights (fairy lights) can provide gentle, distributed lighting that’s bright enough for gentle activity but dim enough to minimise circadian disruption. Choose warm-colored LED string lights rather than white. These work particularly well in bedrooms where you want enough light for your bedtime routine without the overhead light intensity.
Fireplaces, whether wood-burning or gas, provide both warmth and sleep-friendly light. The orange-red spectrum is ideal, and there’s something primally calming about fire. If you have a fireplace, consider making it your primary evening light source in the winter months, using minimal additional lighting.
Gradual reduction is key to all of this. You’re not trying to transition instantly from full brightness to candlelight. You’re stepping down intensity and shifting the colour temperature progressively throughout your evening. Maybe you use normal lighting until 8pm, dim to about 50% brightness and switch to lamps from 8-9pm, and then further dim to minimal lighting with only warm-colored bulbs from 9pm onward. The gradual shift is less disruptive than sudden changes whilst providing clear circadian signals.
Screen Management
Screens are one of the most difficult aspect of evening light management because they’re not just light sources, they’re portals to work, entertainment, social connection, and information. Telling people “don’t use screens after 8pm” is technically correct but practically useless for most people whose lives are deeply integrated with digital technology.
The reality here is that most of you aren’t going to give up evening screen time entirely. Most people use computers for work that extends into evening hours. They watch television or streaming content to wind down. They communicate with friends and family through their phones. They read books on tablets. Complete screen avoidance, while optimal for sleep, is incompatible with how most people actually live. And, having coached many people to better sleep, I know that most people simply won’t give up their screens before bed.
So the question becomes: how do you minimise the circadian disruption of screens while still using them? Here are the strategies, ranked from most to least effective:
1. Reduce total screen time (most effective): Every hour of evening screen time you can eliminate is a win. If you currently watch three hours of TV every evening, reducing to two hours, even without any other interventions, will improve your sleep. Choose one or two shows you genuinely want to watch rather than scrolling until you find something that captures attention. Read physical books instead of tablets. Have conversations instead of messaging. Be brutally honest about what screen time is actually serving you versus what’s just habitual time-filling.
2. Stop all screens at least one hour before bed (highly effective): If you’re going to use screens, at least create a screen-free buffer before sleep. If your bedtime is 10:30pm, no screens after 9:30pm. This gives melatonin an hour to rise before you try to sleep. This single change of just one screen-free hour can transform sleep quality for people who currently use screens right up until they get into bed.
3. Use night mode/night shift features (moderately effective): Every modern device has settings that reduce blue light by shifting screen colour temperature toward red-orange. iOS has Night Shift, Android has Night Light, Windows has Night Light, and Mac has Night Shift. Enable these to activate automatically at sunset or at a set time (I’d recommend 7 or 8pm). The screen will look noticeably more orange, which many people find off-putting initially but adapt to quickly.
These features typically filter wavelengths below 500-550 nanometers, which captures most of the circadian-disrupting blue light. They’re not perfect, and some blue light still gets through, particularly on devices with bright displays, but they reduce the impact substantially compared to unfiltered screens.
4. Reduce screen brightness (moderately effective): Brightness controls are usually easy to access on all devices. Dimming your screen reduces total light exposure even if it doesn’t change the wavelength spectrum. In combination with night mode, this can reduce circadian disruption by 50-70% compared to full-brightness unfiltered screens.
Set your devices to automatically dim in the evening if possible. Many devices can tie brightness to time of day or ambient light levels. You want screens as dim as possible while still being usable.
5. Increase viewing distance (somewhat effective): Light intensity decreases with the square of distance. Holding your phone 30cm from your face delivers four times more light to your eyes than holding it 60cm away. Watching TV from 3 meters delivers far less light than watching from 1.5 meters. This doesn’t change the wavelength issue, but it reduces total light exposure.
This is why television is somewhat less problematic than phones or tablets, as you typically view it from a greater distance, so even though the screen is larger, the light intensity reaching your eyes is lower. Still not ideal, but meaningfully better than a phone screen at arm’s length.
6. Use software solutions (somewhat effective): Applications like f.lux (free) for computers provide more aggressive blue light filtering than built-in night modes and can be customised to your location and schedule. These tools essentially turn your screen quite orange in the evening. The downside is that colour-accurate work (photo editing, design) becomes impossible, but for most evening activities like reading, messaging, or watching videos, the colour distortion is acceptable.
7. Blue light blocking glasses (discussed in the next section): These can be worn while using screens to filter blue light before it reaches your eyes. Effectiveness varies significantly based on the quality of the glasses and how they’re used.
The reality is that you’ll need to combine several of these strategies. Night mode plus reduced brightness plus stopping screens an hour before bed is far more effective than any single intervention. And all of these screen management strategies work better when combined with proper light exposure management throughout the day. Morning light and bright daytime exposure make your circadian system more robust and less easily disrupted by evening screens.
Blue Light Blocking Glasses
Blue light blocking glasses have become popular, marketed as a simple solution to evening screen use. Wear special glasses, block the blue light, keep your circadian rhythm intact, and sleep perfectly. It sounds ideal. The reality is more complicated.
When they work: Blue light blocking glasses can be effective at reducing melatonin suppression from evening light exposure when they actually block the relevant wavelengths, when worn consistently for two to three hours before bed, and when used as part of a comprehensive light exposure management strategy rather than as a standalone solution.
The key specification is that they must block wavelengths below 525 nanometers to be effective. This requires lenses with an orange or red tint (you should be able to see the tint clearly when you hold them up). If the lenses are clear or only slightly yellow, they’re filtering some blue light but not enough to prevent circadian disruption. True circadian-effective glasses make everything look distinctly orange or red.
Research shows that proper blue light blocking glasses can reduce melatonin suppression by 60-90% compared to unfiltered light exposure. They’re particularly useful for people who must use screens or work under bright lights in the hours before bed and have no control over their lighting environment, like shift workers, people who work evening hours, or those who share space with people keeping different schedules.
When they don’t work: If they’re low-quality glasses that don’t actually filter the relevant wavelengths. If you only wear them sporadically or for short periods. If you use them as permission to ignore all other light management strategies. If you take them off repeatedly because they’re uncomfortable, or if you forget to wear them.
The biggest problem with blue light blocking glasses is compliance. They’re uncomfortable for many people, as they add pressure on your nose and ears, they distort colours, making everything look strange, they’re unflattering, they fog up, and they interfere with regular glasses if you wear those. Many people buy them with good intentions, wear them for a few evenings, find them annoying, and abandon them.
Even people who persist often wear them inconsistently. They put them on to watch TV but take them off to check their phone, or wear them some evenings but not others, or put them on at 10pm when they should have started wearing them at 8pm. Inconsistent use dramatically reduces effectiveness.
What to look for: If you’re going to invest in blue light blocking glasses, get ones that actually work:
- Must block all wavelengths below 525nm (check product specifications)
- Should have orange or red tinted lenses; if they’re clear, they’re not blocking enough
- Should fit comfortably enough that you’ll actually wear them for 2-3 hours
- Should fit over your regular glasses if you wear those, or get prescription blue blockers made
- Should have good reviews from people who’ve measured their effectiveness, not just marketing claims
Expect to pay €30-80 for quality blue light blocking glasses. Cheaper options often don’t filter effectively. More expensive options may offer better comfort and style, but aren’t necessarily more effective at wavelength filtering.
The cost-benefit calculation: For most people, reducing evening light through environmental changes (dimmers, lamps, warm bulbs) and reducing screen time is more effective and more sustainable than relying on glasses. Glasses are a useful tool for situations where you cannot control lighting, but they shouldn’t be your primary strategy.
If you work evening shifts under bright lights, if your partner watches TV in bed while you’re trying to sleep, if you have to use screens for work in the evening with no flexibility, these are all situations where blue light blocking glasses might be worth the investment. But if your main issue is that you watch TV and scroll your phone until 10:30pm every night, putting on orange glasses isn’t the solution. Stopping screens at 9:30pm is the solution, and it’s free.
Special Considerations: The Details That Matter
Bathrooms present a unique challenge because you need functional lighting for safety and hygiene, but bathrooms are often used during wind-down routines or in the middle of the night. The solution is layered lighting: keep normal bright lights available for situations requiring visibility (showering, cleaning), but install dim nightlights or path lights for routine evening use and nighttime bathroom trips.
Motion-activated amber nightlights that provide 10-20 lux are ideal for nighttime bathroom use. You can navigate and use the toilet without turning on bright overhead lights that fully wake you and suppress melatonin. These lights are cheap and are easy to install.
Hallways and staircases need enough light for safe navigation, but don’t require full brightness. Motion-activated dim warm-white lights or plug-in nightlights at floor level provide sufficient light for safety whilst minimising circadian disruption.
Reading lights are essential if you read before bed (which is an excellent wind-down activity!). But most reading lights are far too bright and too blue-white in spectrum. Look for reading lights with warm bulbs (2200-2700K), adjustable brightness, and ideally adjustable position so light falls on your book rather than your face. Some specialised book lights clip directly to books and can be positioned to minimise light reaching your eyes while still illuminating the page.
Partner with different schedule: This is one of the most difficult situations. You’re trying to sleep or wind down while your partner is still active, using screens, or wants normal lighting. The solutions require negotiation and compromise: they use headphones for TV or videos, they stay in a different room until they’re ready for bed, they use a headlamp or reading light rather than overhead lights, you use an eye mask, and/or you accept some suboptimal light exposure as the cost of sharing space.
Blackout curtains or dividers can partition shared spaces somewhat. Motion-activated dim path lighting allows the later-schedule person to navigate without turning on bright lights. Smart bulbs that can be controlled individually allow one person to dim lights in their area while the other keeps brighter lights. None of these solutions are perfect, but they’re workable compromises.
Children in the house complicate evening light management because children often need functional lighting later into the evening for homework, reading, or activities, and their bedtime might precede yours. The solution is creating zones: bright lighting in areas where children are active, dimmer lighting in adult spaces. Teaching older children to use task lighting (desk lamps for homework) rather than overhead lights helps. Night lights in children’s rooms should be red or amber, not white or blue.
Light Exposure and Shift Work: Strategic Use
Shift work creates a fundamental conflict with natural circadian rhythms, but strategic light exposure management can mitigate some of the damage. The principle is to use light to trick your circadian system into thinking your work time is day and your sleep time is night, even when the actual sun disagrees.
During your “day” (even if it’s night): Expose yourself to bright light (ideally 1,000+ lux) during your work hours. If you work nights, this might mean using light therapy boxes at your workstation, ensuring work environments are brightly lit, and taking breaks under bright lights. You’re trying to tell your body, “This is daytime, be alert.”
During your “night” (even if it’s day): When it’s time to sleep, you need complete darkness even though it’s biologically daytime. Blackout curtains are super helpful here. Not just room-darkening curtains but actual blackout curtains or shades that eliminate all light. Eye masks can be used to supplement if light leaks through the curtains, and the goal is 0 lux, complete darkness.
If you have to travel home in bright daylight after a night shift, wear sunglasses. Ideally, ones that block blue light heavily. You’re trying to prevent morning light exposure from shifting your circadian rhythm earlier, which would make it harder to sleep when you get home and harder to be alert for your next shift.
Light timing to adjust schedule: When your schedule changes (i.e. rotating from day shifts to night shifts), use light exposure strategically to help your circadian rhythm adapt. If you’re shifting later (moving from day to evening shifts), expose yourself to bright light in the evening and avoid morning light. If you’re shifting earlier, expose yourself to bright morning light and avoid evening light.
This doesn’t make shift work easy on your body, and the health costs of circadian disruption persist regardless of how well you manage light. But strategic light exposure management can reduce the impact and make the transitions less brutal.
Measuring Light
One reason light exposure management is difficult is that human perception of brightness is logarithmic and highly context-dependent. What feels like “dim lighting” might be 200 lux, which is far too bright for optimal evening light. What feels like “adequate morning light” indoors might be 500 lux, which is far too dim for circadian entrainment.
You can download free lux meter apps for your smartphone that use your camera to measure light intensity. They’re not laboratory-accurate, but they’re precise enough for practical purposes. Measuring your actual light exposure often reveals surprising findings: your “bright” office is only 400 lux. Your “dim” evening lighting is 300 lux. Your morning bedroom, where you thought you were getting good light through the window, is only 200 lux.
What different light levels look like:
- 0-10 lux: Very dim, candlelight, small nightlights
- 10-50 lux: Dim, enough to navigate but not to read comfortably
- 50-200 lux: Moderate, enough for most activities but still fairly dim
- 200-500 lux: Typical indoor lighting, comfortable for reading and work
- 500-1,000 lux: Bright indoor lighting, well-lit office
- 1,000-10,000 lux: Very bright, outdoor shade or overcast day
- 10,000+ lux: Outdoor daylight
Once you start measuring, you develop intuition about light levels and can make better decisions about your environment. You realise that “going near a window” provides maybe 500 lux, while actually going outside provides 20,000+ lux. You realise your evening lighting that feels dim is still 250 lux, which is far too bright for optimal melatonin production.
Troubleshooting Common Light Exposure Management Obstacles
“I work indoors all day and can’t get outside.” Take breaks. Even five-minute breaks outside three times daily provides substantial light exposure. Lunch outdoors is non-negotiable if you’re serious about light exposure management. Position your workspace near windows. Consider using a light therapy box at your desk during morning hours if outdoor access is genuinely impossible.
The reality is that some jobs make this extremely difficult. I have worked with surgeons in operating rooms, factory workers, and people in basement offices, who all find it incredibly difficult to actually get the right light exposure. If you literally cannot get outdoor light during work hours, you need to compensate with extended morning light exposure (30-60 minutes before work), light therapy at your workspace if possible, and aggressive evening light reduction to create maximum day-night contrast despite reduced daytime light.
“I need screens for work at night.” If work genuinely requires screen use during your wind-down hours, implement every possible mitigation: night mode enabled and set to maximum filtering, screen brightness as low as possible, blue light blocking glasses, take frequent breaks to close your eyes or look away, stop screens at least thirty minutes before bed, even if you were working until late.
Also, question whether the screen use is genuinely necessary or whether it’s a habit you could change. Can some work be done earlier in the day? Can you set boundaries around evening work except in true emergencies? Can you use paper and pen for some tasks that currently happen on screens?
“My partner watches TV in bed.” This is genuinely difficult and requires explicit negotiation. Options: they use headphones, and you use an eye mask, they watch in another room until you’re asleep, you go to bed earlier and sleep through it, you compromise on screen brightness and viewing time, or you acknowledge that this is damaging your sleep and something needs to change.
Some relationships require choosing between optimal sleep and time together in the evening. That’s a legitimate trade-off, but it should be a conscious choice rather than an unexamined pattern. Maybe two or three nights per week you prioritise sleep, and they watch TV elsewhere. Maybe you share screen time until 9pm, and then you diverge in your routines. Maybe you invest in blackout sleep masks and accept some compromise.
“I wake to pee and turn on bright bathroom lights.” Install motion-activated dim amber nightlights in the bathroom and hallway. These provide adequate light for safe navigation and bathroom use whilst being dim enough (10-20 lux) that they won’t fully wake you or suppress melatonin. The difference in how easily you return to sleep after using a dim red nightlight versus bright bathroom lights is remarkable.
Light Exposure Management Conclusion
Light exposure management isn’t about becoming neurotic about every photon entering your eyes. It’s about recognising that your biology evolved in an environment with strong day-night light contrast, with tens of thousands of lux during the day, zero lux at night, and that modern life has completely obliterated this contrast. You’re now living in a perpetual twilight: too dim during the day, too bright at night.
This confusion at the level of your circadian clock cascades through every system in your body. Your sleep suffers most obviously, but so does your metabolism, immune function, mood, cognitive performance, and long-term health. The research linking circadian disruption to obesity, diabetes, cardiovascular disease, depression, and even cancer is robust and growing.
When you implement proper light exposure management (bright morning light, adequate daytime light, progressively dim evening light), you’re not just improving sleep. You’re restoring the fundamental signal your body uses to coordinate thousands of biological processes. You’re giving your circadian system the clarity it needs to do its job, which is regulating the rhythm of your entire physiology.
This is why I’m asking you to take this seriously despite how inconvenient it is. Despite the fact that you’ll need to change habits, modify your environment, potentially invest in new lighting, and set boundaries around evening activities. Despite the social awkwardness of leaving gatherings earlier to protect your evening light exposure, or declining invitations to late-evening events.
You’re choosing to work with your biology rather than against it. You’re acknowledging that you’re not separate from nature, not somehow exempt from the constraints that govern all living things. You’re a diurnal species, adapted to be active during light and rest during darkness. No amount of artificial light makes you something else. You can override these patterns temporarily, but you pay the cost in depleted well-being and compromised health.
The modern world makes optimal light exposure difficult. It’s designed around productivity and convenience, not circadian health. But you have more control than you think. You can go outside in the morning. You can dim your lights in the evening. You can stop using screens an hour before bed. These aren’t monumental changes. They’re small adjustments that compound into transformed sleep and enhanced capacity for living well.
So, with that in mind, here’s your implementation plan:
Tomorrow, set your alarm fifteen minutes earlier than usual. When it goes off, get up immediately and go outside. Spend ten to fifteen minutes in outdoor light; walk, stand, sit, drink coffee, check your phone if you must, but be outside in natural light. Do this every day for a week.
After one week, add evening light reduction: two hours before your bedtime, dim all lights to 50% or switch to lamps only. One hour before bedtime, dim the lights further to minimal lighting. Notice how you feel.
After two weeks, address screens: stop all screen use one hour before your target bedtime. Read a physical book, journal, stretch, talk to your partner, do your wind-down routine; anything but screens.
Implement gradually. Master morning light before adding evening restrictions. Build the habits layer by layer rather than trying to implement everything at once, failing, and giving up.
Your circadian rhythm wants to work for you. It’s an ancient, powerful system that knows exactly when you should be alert and when you should sleep. But it needs clear signals. Give it those signals through proper light exposure management, and watch how much easier everything becomes.
Start tomorrow morning. Go outside within thirty minutes of waking. Everything else follows from there.
As with everything, there is always more to learn, and we haven’t even begun to scratch the surface with all this stuff. However, if you are interested in staying up to date with all our content, we recommend subscribing to our newsletter and bookmarking our free content page. We do have a lot of content on sleep in our sleep hub.
The previous article in this series was Wind Down Routine: Foundational Sleep Hygiene, and the next article in this series is Sleep Environment Optimisation: Foundational Sleep Hygiene.
If you would like more help with your training (or nutrition), we do also have online coaching spaces available.
We also recommend reading our foundational nutrition articles, along with our foundational articles on exercise and stress management, if you really want to learn more about how to optimise your lifestyle. If you want even more free information on sleep, you can follow us on Instagram, YouTube or listen to the podcast, where we discuss all the little intricacies of exercise.
Finally, if you want to learn how to coach nutrition, then consider our Nutrition Coach Certification course. We do also have an exercise program design course, if you are a coach who wants to learn more about effective program design and how to coach it. We do have other courses available too, notably as a sleep course. If you don’t understand something, or you just need clarification, you can always reach out to us on Instagram or via email.
References and Further Reading
Vyazovskiy, V. (2015). Sleep, recovery, and metaregulation: explaining the benefits of sleep. Nature and Science of Sleep, 171. http://doi.org/10.2147/nss.s54036
Sharma, S., & Kavuru, M. (2010). Sleep and Metabolism: An Overview. International Journal of Endocrinology, 2010, 1–12. http://doi.org/10.1155/2010/270832
Yoo, S.-S., Gujar, N., Hu, P., Jolesz, F. A., & Walker, M. P. (2007). The human emotional brain without sleep — a prefrontal amygdala disconnect. Current Biology, 17(20). http://doi.org/10.1016/j.cub.2007.08.007
Copinschi G. Metabolic and endocrine effects of sleep deprivation. Essent Psychopharmacol. 2005;6(6):341-7. PMID: 16459757. https://pubmed.ncbi.nlm.nih.gov/16459757/
Spiegel, K., Leproult, R., L’Hermite-Balériaux, M., Copinschi, G., Penev, P. D., & Cauter, E. V. (2004). Leptin Levels Are Dependent on Sleep Duration: Relationships with Sympathovagal Balance, Carbohydrate Regulation, Cortisol, and Thyrotropin. The Journal of Clinical Endocrinology & Metabolism, 89(11), 5762–5771. http://doi.org/10.1210/jc.2004-1003
Nedeltcheva, A. V., Kilkus, J. M., Imperial, J., Kasza, K., Schoeller, D. A., & Penev, P. D. (2008). Sleep curtailment is accompanied by increased intake of calories from snacks. The American Journal of Clinical Nutrition, 89(1), 126–133. http://doi.org/10.3945/ajcn.2008.26574
Mullington, J. M., Chan, J. L., Dongen, H. P. A. V., Szuba, M. P., Samaras, J., Price, N. J., … Mantzoros, C. S. (2003). Sleep Loss Reduces Diurnal Rhythm Amplitude of Leptin in Healthy Men. Journal of Neuroendocrinology, 15(9), 851–854. http://doi.org/10.1046/j.1365-2826.2003.01069.x
Leproult, R., & Cauter, E. V. (2009). Role of Sleep and Sleep Loss in Hormonal Release and Metabolism. Pediatric Neuroendocrinology Endocrine Development, 11–21. http://doi.org/10.1159/000262524
Spaeth, A. M., Dinges, D. F., & Goel, N. (2013). Effects of Experimental Sleep Restriction on Weight Gain, Caloric Intake, and Meal Timing in Healthy Adults. Sleep, 36(7), 981–990. http://doi.org/10.5665/sleep.2792
Calvin, A. D., Carter, R. E., Adachi, T., Macedo, P. G., Albuquerque, F. N., Walt, C. V. D., … Somers, V. K. (2013). Effects of Experimental Sleep Restriction on Caloric Intake and Activity Energy Expenditure. Chest, 144(1), 79–86. http://doi.org/10.1378/chest.12-2829
Markwald, R. R., Melanson, E. L., Smith, M. R., Higgins, J., Perreault, L., Eckel, R. H., & Wright, K. P. (2013). Impact of insufficient sleep on total daily energy expenditure, food intake, and weight gain. Proceedings of the National Academy of Sciences, 110(14), 5695–5700. http://doi.org/10.1073/pnas.1216951110
Cauter, E. V., Spiegel, K., Tasali, E., & Leproult, R. (2008). Metabolic consequences of sleep and sleep loss. Sleep Medicine, 9. http://doi.org/10.1016/s1389-9457(08)70013-3
Spiegel, K., Leproult, R., & Cauter, E. V. (1999). Impact of sleep debt on metabolic and endocrine function. The Lancet, 354(9188), 1435–1439. http://doi.org/10.1016/s0140-6736(99)01376-8
Ness, K. M., Strayer, S. M., Nahmod, N. G., Schade, M. M., Chang, A.-M., Shearer, G. C., & Buxton, O. M. (2019). Four nights of sleep restriction suppress the postprandial lipemic response and decrease satiety. Journal of Lipid Research, 60(11), 1935–1945. http://doi.org/10.1194/jlr.p094375
Hirotsu, C., Tufik, S., & Andersen, M. L. (2015). Interactions between sleep, stress, and metabolism: From physiological to pathological conditions. Sleep Science, 8(3), 143–152. http://doi.org/10.1016/j.slsci.2015.09.002
Morselli, L., Leproult, R., Balbo, M., & Spiegel, K. (2010). Role of sleep duration in the regulation of glucose metabolism and appetite. Best Practice & Research Clinical Endocrinology & Metabolism, 24(5), 687–702. http://doi.org/10.1016/j.beem.2010.07.005
Lamon, S., Morabito, A., Arentson-Lantz, E., Knowles, O., Vincent, G. E., Condo, D., … Aisbett, B. (2020). The effect of acute sleep deprivation on skeletal muscle protein synthesis and the hormonal environment. http://doi.org/10.1101/2020.03.09.984666
Lipton, J. O., & Sahin, M. (2014). The Neurology of mTOR. Neuron, 84(2), 275–291. http://doi.org/10.1016/j.neuron.2014.09.034
Tudor, J. C., Davis, E. J., Peixoto, L., Wimmer, M. E., Tilborg, E. V., Park, A. J., … Abel, T. (2016). Sleep deprivation impairs memory by attenuating mTORC1-dependent protein synthesis. Science Signaling, 9(425). http://doi.org/10.1126/scisignal.aad4949
Dattilo, M., Antunes, H., Medeiros, A., Neto, M. M., Souza, H., Tufik, S., & Mello, M. D. (2011). Sleep and muscle recovery: Endocrinological and molecular basis for a new and promising hypothesis. Medical Hypotheses, 77(2), 220–222. http://doi.org/10.1016/j.mehy.2011.04.017
Thornton, S. N., & Trabalon, M. (2014). Chronic dehydration is associated with obstructive sleep apnoea syndrome. Clinical Science, 128(3), 225–225. http://doi.org/10.1042/cs20140496
Rosinger, A. Y., Chang, A.-M., Buxton, O. M., Li, J., Wu, S., & Gao, X. (2018). Short sleep duration is associated with inadequate hydration: cross-cultural evidence from US and Chinese adults. Sleep, 42(2). http://doi.org/10.1093/sleep/zsy210
Watson, A. M. (2017). Sleep and Athletic Performance. Current Sports Medicine Reports, 16(6), 413–418. http://doi.org/10.1249/jsr.0000000000000418
Bonnar, D., Bartel, K., Kakoschke, N., & Lang, C. (2018). Sleep Interventions Designed to Improve Athletic Performance and Recovery: A Systematic Review of Current Approaches. Sports Medicine, 48(3), 683–703. http://doi.org/10.1007/s40279-017-0832-x
Saidi, O., Davenne, D., Lehorgne, C., & Duché, P. (2020). Effects of timing of moderate exercise in the evening on sleep and subsequent dietary intake in lean, young, healthy adults: randomized crossover study. European Journal of Applied Physiology, 120(7), 1551–1562. http://doi.org/10.1007/s00421-020-04386-6
Abedelmalek, S., Chtourou, H., Aloui, A., Aouichaoui, C., Souissi, N., & Tabka, Z. (2012). Effect of time of day and partial sleep deprivation on plasma concentrations of IL-6 during a short-term maximal performance. European Journal of Applied Physiology, 113(1), 241–248. http://doi.org/10.1007/s00421-012-2432-7
Azboy, O., & Kaygisiz, Z. (2009). Effects of sleep deprivation on cardiorespiratory functions of the runners and volleyball players during rest and exercise. Acta Physiologica Hungarica, 96(1), 29–36. http://doi.org/10.1556/aphysiol.96.2009.1.3
Bird, S. P. (2013). Sleep, Recovery, and Athletic Performance. Strength and Conditioning Journal, 35(5), 43–47. http://doi.org/10.1519/ssc.0b013e3182a62e2f
Blumert, P. A., Crum, A. J., Ernsting, M., Volek, J. S., Hollander, D. B., Haff, E. E., & Haff, G. G. (2007). The Acute Effects of Twenty-Four Hours of Sleep Loss on the Performance of National-Caliber Male Collegiate Weightlifters. The Journal of Strength and Conditioning Research, 21(4), 1146. http://doi.org/10.1519/r-21606.1
Chase, J. D., Roberson, P. A., Saunders, M. J., Hargens, T. A., Womack, C. J., & Luden, N. D. (2017). One night of sleep restriction following heavy exercise impairs 3-km cycling time-trial performance in the morning. Applied Physiology, Nutrition, and Metabolism, 42(9), 909–915. http://doi.org/10.1139/apnm-2016-0698
Edwards, B. J., & Waterhouse, J. (2009). Effects of One Night of Partial Sleep Deprivation upon Diurnal Rhythms of Accuracy and Consistency in Throwing Darts. Chronobiology International, 26(4), 756–768. http://doi.org/10.1080/07420520902929037
Fullagar, H. H. K., Skorski, S., Duffield, R., Hammes, D., Coutts, A. J., & Meyer, T. (2014). Sleep and Athletic Performance: The Effects of Sleep Loss on Exercise Performance, and Physiological and Cognitive Responses to Exercise. Sports Medicine, 45(2), 161–186. http://doi.org/10.1007/s40279-014-0260-0
Gupta, L., Morgan, K., & Gilchrist, S. (2016). Does Elite Sport Degrade Sleep Quality? A Systematic Review. Sports Medicine, 47(7), 1317–1333. http://doi.org/10.1007/s40279-016-0650-6
Hausswirth, C., Louis, J., Aubry, A., Bonnet, G., Duffield, R., & Meur, Y. L. (2014). Evidence of Disturbed Sleep and Increased Illness in Overreached Endurance Athletes. Medicine & Science in Sports & Exercise, 46(5), 1036–1045. http://doi.org/10.1249/mss.0000000000000177
Mah, C. D., Mah, K. E., Kezirian, E. J., & Dement, W. C. (2011). The Effects of Sleep Extension on the Athletic Performance of Collegiate Basketball Players. Sleep, 34(7), 943–950. http://doi.org/10.5665/sleep.1132
Milewski, M. D., Skaggs, D. L., Bishop, G. A., Pace, J. L., Ibrahim, D. A., Wren, T. A., & Barzdukas, A. (2014). Chronic Lack of Sleep is Associated With Increased Sports Injuries in Adolescent Athletes. Journal of Pediatric Orthopaedics, 34(2), 129–133. http://doi.org/10.1097/bpo.0000000000000151
Mougin, F., Bourdin, H., Simon-Rigaud, M., Didier, J., Toubin, G., & Kantelip, J. (1996). Effects of a Selective Sleep Deprivation on Subsequent Anaerobic Performance. International Journal of Sports Medicine, 17(02), 115–119. http://doi.org/10.1055/s-2007-972818
Oliver, S. J., Costa, R. J. S., Laing, S. J., Bilzon, J. L. J., & Walsh, N. P. (2009). One night of sleep deprivation decreases treadmill endurance performance. European Journal of Applied Physiology, 107(2), 155–161. http://doi.org/10.1007/s00421-009-1103-9
Pallesen, S., Gundersen, H. S., Kristoffersen, M., Bjorvatn, B., Thun, E., & Harris, A. (2017). The Effects of Sleep Deprivation on Soccer Skills. Perceptual and Motor Skills, 124(4), 812–829. http://doi.org/10.1177/0031512517707412
Reilly, T., & Piercy, M. (1994). The effect of partial sleep deprivation on weight-lifting performance. Ergonomics, 37(1), 107–115. http://doi.org/10.1080/00140139408963628
Rossa, K. R., Smith, S. S., Allan, A. C., & Sullivan, K. A. (2014). The Effects of Sleep Restriction on Executive Inhibitory Control and Affect in Young Adults. Journal of Adolescent Health, 55(2), 287–292. http://doi.org/10.1016/j.jadohealth.2013.12.034
Sargent, C., & Roach, G. D. (2016). Sleep duration is reduced in elite athletes following night-time competition. Chronobiology International, 33(6), 667–670. http://doi.org/10.3109/07420528.2016.1167715
Skein, M., Duffield, R., Edge, J., Short, M. J., & Mündel, T. (2011). Intermittent-Sprint Performance and Muscle Glycogen after 30 h of Sleep Deprivation. Medicine & Science in Sports & Exercise, 43(7), 1301–1311. http://doi.org/10.1249/mss.0b013e31820abc5a
Souissi, N., Sesboüé, B., Gauthier, A., Larue, J., & Davenne, D. (2003). Effects of one nights sleep deprivation on anaerobic performance the following day. European Journal of Applied Physiology, 89(3), 359–366. http://doi.org/10.1007/s00421-003-0793-7
Caia, J., Kelly, V. G., & Halson, S. L. (2017). The role of sleep in maximising performance in elite athletes. Sport, Recovery, and Performance, 151–167. http://doi.org/10.4324/9781315268149-11
Alley, J. R., Mazzochi, J. W., Smith, C. J., Morris, D. M., & Collier, S. R. (2015). Effects of Resistance Exercise Timing on Sleep Architecture and Nocturnal Blood Pressure. Journal of Strength and Conditioning Research, 29(5), 1378–1385. http://doi.org/10.1519/jsc.0000000000000750
Kovacevic, A., Mavros, Y., Heisz, J. J., & Singh, M. A. F. (2018). The effect of resistance exercise on sleep: A systematic review of randomized controlled trials. Sleep Medicine Reviews, 39, 52–68. http://doi.org/10.1016/j.smrv.2017.07.002
Herrick, J. E., Puri, S., & Richards, K. C. (2017). Resistance training does not alter same-day sleep architecture in institutionalized older adults. Journal of Sleep Research, 27(4). http://doi.org/10.1111/jsr.12590
Edinger, J. D., Morey, M. C., Sullivan, R. J., Higginbotham, M. B., Marsh, G. R., Dailey, D. S., & McCall, W. V. (1993). Aerobic fitness, acute exercise and sleep in older men. Sleep, 16(4), 351-359. https://doi.org/10.1093/sleep/16.4.351
King, A. C. (1997). Moderate-intensity exercise and self-rated quality of sleep in older adults. A randomized controlled trial. JAMA: The Journal of the American Medical Association, 277(1), 32–37. http://doi.org/10.1001/jama.277.1.32
Passos, G. S., Poyares, D., Santana, M. G., Garbuio, S. A., Tufik, S., & Mello, M. T. (2010). Effect of Acute Physical Exercise on Patients with Chronic Primary Insomnia. Journal of Clinical Sleep Medicine, 06(03), 270–275. http://doi.org/10.5664/jcsm.27825
Reid, K. J., Baron, K. G., Lu, B., Naylor, E., Wolfe, L., & Zee, P. C. (2010). Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Medicine, 11(9), 934–940. http://doi.org/10.1016/j.sleep.2010.04.014
Viana, V. A. R., Esteves, A. M., Boscolo, R. A., Grassmann, V., Santana, M. G., Tufik, S., & Mello, M. T. D. (2011). The effects of a session of resistance training on sleep patterns in the elderly. European Journal of Applied Physiology, 112(7), 2403–2408. http://doi.org/10.1007/s00421-011-2219-2
Herring, M., Kline, C., & Oconnor, P. (2015). Effects of Exercise Training On Self-reported Sleep Among Young Women with Generalized Anxiety Disorder (GAD). European Psychiatry, 30, 465. http://doi.org/10.1016/s0924-9338(15)31893-9
Kredlow, M. A., Capozzoli, M. C., Hearon, B. A., Calkins, A. W., & Otto, M. W. (2015). The effects of physical activity on sleep: a meta-analytic review. Journal of Behavioral Medicine, 38(3), 427–449. http://doi.org/10.1007/s10865-015-9617-6
Yang, P.-Y., Ho, K.-H., Chen, H.-C., & Chien, M.-Y. (2012). Exercise training improves sleep quality in middle-aged and older adults with sleep problems: a systematic review. Journal of Physiotherapy, 58(3), 157–163. http://doi.org/10.1016/s1836-9553(12)70106-6
Kline, C. E., Sui, X., Hall, M. H., Youngstedt, S. D., Blair, S. N., Earnest, C. P., & Church, T. S. (2012). Dose–response effects of exercise training on the subjective sleep quality of postmenopausal women: exploratory analyses of a randomised controlled trial. BMJ Open, 2(4). http://doi.org/10.1136/bmjopen-2012-001044
Fairbrother, K., Cartner, B. W., Triplett, N., Morris, D. M., & Collier, S. R. (2011). The Effects of Aerobic Exercise Timing on Sleep Architecture. Medicine & Science in Sports & Exercise, 43(Suppl 1), 879. http://doi.org/10.1249/01.mss.0000402452.16375.20
Youngstedt, S. D., & Kline, C. E. (2006). Epidemiology of exercise and sleep. Sleep and Biological Rhythms, 4(3), 215–221. http://doi.org/10.1111/j.1479-8425.2006.00235.x
Stenholm, S., Head, J., Kivimäki, M., Hanson, L. L. M., Pentti, J., Rod, N. H., … Vahtera, J. (2018). Sleep Duration and Sleep Disturbances as Predictors of Healthy and Chronic Disease–Free Life Expectancy Between Ages 50 and 75: A Pooled Analysis of Three Cohorts. The Journals of Gerontology: Series A, 74(2), 204–210. http://doi.org/10.1093/gerona/gly01
Xiao, Q., Keadle, S. K., Hollenbeck, A. R., & Matthews, C. E. (2014). Sleep Duration and Total and Cause-Specific Mortality in a Large US Cohort: Interrelationships With Physical Activity, Sedentary Behavior, and Body Mass Index. American Journal of Epidemiology, 180(10), 997–1006. http://doi.org/10.1093/aje/kwu222
Reynolds, A. C., Dorrian, J., Liu, P. Y., Dongen, H. P. A. V., Wittert, G. A., Harmer, L. J., & Banks, S. (2012). Impact of Five Nights of Sleep Restriction on Glucose Metabolism, Leptin and Testosterone in Young Adult Men. PLoS ONE, 7(7). http://doi.org/10.1371/journal.pone.0041218
Åkerstedt, T., Palmblad, J., Torre, B. D. L., Marana, R., & Gillberg, M. (1980). Adrenocortical and Gonadal Steroids During Sleep Deprivation. Sleep, 3(1), 23–30. http://doi.org/10.1093/sleep/3.1.23
Cortés-Gallegos, V., Castañeda, G., Alonso, R., Sojo, I., Carranco, A., Cervantes, C., & Parra, A. (1983). Sleep Deprivation Reduces Circulating Androgens in Healthy Men. Archives of Andrology, 10(1), 33–37. http://doi.org/10.3109/01485018308990167
González-Santos, M. R., Gajá-Rodíguez, O. V., Alonso-Uriarte, R., Sojo-Aranda, I., & Cortés-Gallegos, V. (1989). Sleep Deprivation and Adaptive Hormonal Responses of Healthy Men. Archives of Andrology, 22(3), 203–207. http://doi.org/10.3109/01485018908986773
Penev, P. D. (2007). Association Between Sleep and Morning Testosterone Levels In Older Men. Sleep, 30(4), 427–432. http://doi.org/10.1093/sleep/30.4.427
Kloss, J. D., Perlis, M. L., Zamzow, J. A., Culnan, E. J., & Gracia, C. R. (2015). Sleep, sleep disturbance, and fertility in women. Sleep Medicine Reviews, 22, 78–87. http://doi.org/10.1016/j.smrv.2014.10.005
Mahoney, M. M. (2010). Shift Work, Jet Lag, and Female Reproduction. International Journal of Endocrinology, 2010, 1–9. http://doi.org/10.1155/2010/813764
Labyak, S., Lava, S., Turek, F., & Zee, P. (2002). Effects Of Shiftwork On Sleep And Menstrual Function In Nurses. Health Care for Women International, 23(6-7), 703–714. http://doi.org/10.1080/07399330290107449
Pal, L., Bevilacqua, K., Zeitlian, G., Shu, J., & Santoro, N. (2008). Implications of diminished ovarian reserve (DOR) extend well beyond reproductive concerns. Menopause, 15(6), 1086–1094. http://doi.org/10.1097/gme.0b013e3181728467
Axelsson, G., Rylander, R., & Molin, I. (1989). Outcome of pregnancy in relation to irregular and inconvenient work schedules. Occupational and Environmental Medicine, 46(6), 393–398. http://doi.org/10.1136/oem.46.6.393
Bisanti, L., Olsen, J., Basso, O., Thonneau, P., & Karmaus, W. (1996). Shift Work and Subfecundity: A European Multicenter Study. Journal of Occupational & Environmental Medicine, 38(4), 352–358. http://doi.org/10.1097/00043764-199604000-00012
Rossmanith, W. G. (1998). The impact of sleep on gonadotropin secretion. Gynecological Endocrinology, 12(6), 381–389. http://doi.org/10.3109/09513599809012840
Fernando, S., & Rombauts, L. (2014). Melatonin: shedding light on infertility? – a review of the recent literature. Journal of Ovarian Research, 7(1). http://doi.org/10.1186/s13048-014-0098-y
Rocha, C., Rato, L., Martins, A., Alves, M., & Oliveira, P. (2015). Melatonin and Male Reproductive Health: Relevance of Darkness and Antioxidant Properties. Current Molecular Medicine, 15(4), 299–311. http://doi.org/10.2174/1566524015666150505155530
Song, C., Peng, W., Yin, S., Zhao, J., Fu, B., Zhang, J., … Zhang, Y. (2016). Melatonin improves age-induced fertility decline and attenuates ovarian mitochondrial oxidative stress in mice. Scientific Reports, 6(1). http://doi.org/10.1038/srep35165
Espino, J., Macedo, M., Lozano, G., Ortiz, Á., Rodríguez, C., Rodríguez, A. B., & Bejarano, I. (2019). Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants, 8(9), 338. http://doi.org/10.3390/antiox8090338
Tamura, H., Takasaki, A., Taketani, T., Tanabe, M., Kizuka, F., Lee, L., … Sugino, N. (2012). The role of melatonin as an antioxidant in the follicle. Journal of Ovarian Research, 5(1), 5. http://doi.org/10.1186/1757-2215-5-5
Saaresranta, T., & Polo, O. (2003). Sleep-disordered breathing and hormones. European Respiratory Journal, 22(1), 161–172. http://doi.org/10.1183/09031936.03.00062403
Cappuccio, F. P., Cooper, D., Delia, L., Strazzullo, P., & Miller, M. A. (2011). Sleep duration predicts cardiovascular outcomes: a systematic review and meta-analysis of prospective studies. European Heart Journal, 32(12), 1484–1492. http://doi.org/10.1093/eurheartj/ehr007
Jansen, E. C., Dunietz, G. L., Tsimpanouli, M.-E., Guyer, H. M., Shannon, C., Hershner, S. D., … Baylin, A. (2018). Sleep, Diet, and Cardiometabolic Health Investigations: a Systematic Review of Analytic Strategies. Current Nutrition Reports, 7(4), 235–258. http://doi.org/10.1007/s13668-018-0240-3
Knutson, K. L., Cauter, E. V., Rathouz, P. J., Yan, L. L., Hulley, S. B., Liu, K., & Lauderdale, D. S. (2009). Association Between Sleep and Blood Pressure in Midlife. Archives of Internal Medicine, 169(11), 1055. http://doi.org/10.1001/archinternmed.2009.119
Besedovsky, L., Lange, T., & Born, J. (2011). Sleep and immune function. Pflügers Archiv – European Journal of Physiology, 463(1), 121–137. http://doi.org/10.1007/s00424-011-1044-0
Besedovsky, L., Lange, T., & Haack, M. (2019). The Sleep-Immune Crosstalk in Health and Disease. Physiological Reviews, 99(3), 1325–1380. http://doi.org/10.1152/physrev.00010.2018
Orr, W. C., Fass, R., Sundaram, S. S., & Scheimann, A. O. (2020). The effect of sleep on gastrointestinal functioning in common digestive diseases. The Lancet Gastroenterology & Hepatology, 5(6), 616–624. http://doi.org/10.1016/s2468-1253(19)30412-1
Tang, Y., Preuss, F., Turek, F. W., Jakate, S., & Keshavarzian, A. (2009). Sleep deprivation worsens inflammation and delays recovery in a mouse model of colitis. Sleep Medicine, 10(6), 597–603. http://doi.org/10.1016/j.sleep.2008.12.009
Chen, Y., Tan, F., Wei, L., Li, X., Lyu, Z., Feng, X., … Li, N. (2018). Sleep duration and the risk of cancer: a systematic review and meta-analysis including dose–response relationship. BMC Cancer, 18(1). http://doi.org/10.1186/s12885-018-5025-y
Almendros, I., Martinez-Garcia, M. A., Farré, R., & Gozal, D. (2020). Obesity, sleep apnea, and cancer. International Journal of Obesity, 44(8), 1653–1667. http://doi.org/10.1038/s41366-020-0549-z
Erren, T. C., Falaturi, P., Morfeld, P., Knauth, P., Reiter, R. J., & Piekarski, C. (2010). Shift Work and Cancer. Deutsches Aerzteblatt Online. http://doi.org/10.3238/arztebl.2010.0657
Bernert, R. A., Kim, J. S., Iwata, N. G., & Perlis, M. L. (2015). Sleep Disturbances as an Evidence-Based Suicide Risk Factor. Current Psychiatry Reports, 17(3). http://doi.org/10.1007/s11920-015-0554-4
Kim, J.-H., Park, E.-C., Cho, W.-H., Park, J.-Y., Choi, W.-J., & Chang, H.-S. (2013). Association between Total Sleep Duration and Suicidal Ideation among the Korean General Adult Population. Sleep, 36(10), 1563–1572. http://doi.org/10.5665/sleep.3058
Mccall, W. V., & Black, C. G. (2013). The Link Between Suicide and Insomnia: Theoretical Mechanisms. Current Psychiatry Reports, 15(9). http://doi.org/10.1007/s11920-013-0389-9
Li, S. X., Lam, S. P., Zhang, J., Yu, M. W. M., Chan, J. W. Y., Chan, C. S. Y., … Wing, Y.-K. (2016). Sleep Disturbances and Suicide Risk in an 8-Year Longitudinal Study of Schizophrenia-Spectrum Disorders. Sleep, 39(6), 1275–1282. http://doi.org/10.5665/sleep.5852
Littlewood, D. L., Gooding, P., Kyle, S. D., Pratt, D., & Peters, S. (2016). Understanding the role of sleep in suicide risk: qualitative interview study. BMJ Open, 6(8). http://doi.org/10.1136/bmjopen-2016-012113
Lin, H.-T., Lai, C.-H., Perng, H.-J., Chung, C.-H., Wang, C.-C., Chen, W.-L., & Chien, W.-C. (2018). Insomnia as an independent predictor of suicide attempts: a nationwide population-based retrospective cohort study. BMC Psychiatry, 18(1). http://doi.org/10.1186/s12888-018-1702-2
Freeman, D., Sheaves, B., Waite, F., Harvey, A. G., & Harrison, P. J. (2020). Sleep disturbance and psychiatric disorders. The Lancet Psychiatry, 7(7), 628–637. http://doi.org/10.1016/s2215-0366(20)30136-x
Benca, R. M. (1992). Sleep and Psychiatric Disorders. Archives of General Psychiatry, 49(8), 651. http://doi.org/10.1001/archpsyc.1992.01820080059010
Breslau, N., Roth, T., Rosenthal, L., & Andreski, P. (1996). Sleep disturbance and psychiatric disorders: A longitudinal epidemiological study of young Adults. Biological Psychiatry, 39(6), 411–418. http://doi.org/10.1016/0006-3223(95)00188-3
Baglioni, C., Nanovska, S., Regen, W., Spiegelhalder, K., Feige, B., Nissen, C., … Riemann, D. (2016). Sleep and mental disorders: A meta-analysis of polysomnographic research. Psychological Bulletin, 142(9), 969–990. http://doi.org/10.1037/bul0000053
Goldstein, A. N., & Walker, M. P. (2014). The Role of Sleep in Emotional Brain Function. Annual Review of Clinical Psychology, 10(1), 679–708. http://doi.org/10.1146/annurev-clinpsy-032813-153716
Postuma, R. B., Iranzo, A., Hu, M., Högl, B., Boeve, B. F., Manni, R., … Pelletier, A. (2019). Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain, 142(3), 744–759. http://doi.org/10.1093/brain/awz030
Wintler, T., Schoch, H., Frank, M. G., & Peixoto, L. (2020). Sleep, brain development, and autism spectrum disorders: Insights from animal models. Journal of Neuroscience Research, 98(6), 1137–1149. http://doi.org/10.1002/jnr.24619
Shokri-Kojori, E., Wang, G.-J., Wiers, C. E., Demiral, S. B., Guo, M., Kim, S. W., … Volkow, N. D. (2018). β-Amyloid accumulation in the human brain after one night of sleep deprivation. Proceedings of the National Academy of Sciences, 115(17), 4483–4488. http://doi.org/10.1073/pnas.1721694115
Mantovani, S., Smith, S. S., Gordon, R., & Osullivan, J. D. (2018). An overview of sleep and circadian dysfunction in Parkinsons disease. Journal of Sleep Research, 27(3). http://doi.org/10.1111/jsr.12673
Malhotra, R. K. (2018). Neurodegenerative Disorders and Sleep. Sleep Medicine Clinics, 13(1), 63–70. http://doi.org/10.1016/j.jsmc.2017.09.006
Huang, L.-B., Tsai, M.-C., Chen, C.-Y., & Hsu, S.-C. (2013). The Effectiveness of Light/Dark Exposure to Treat Insomnia in Female Nurses Undertaking Shift Work during the Evening/Night Shift. Journal of Clinical Sleep Medicine, 09(07), 641–646. http://doi.org/10.5664/jcsm.2824
Zhang, Y., & Papantoniou, K. (2019). Night shift work and its carcinogenicity. The Lancet Oncology, 20(10). http://doi.org/10.1016/s1470-2045(19)30578-9
Perry-Jenkins, M., Goldberg, A. E., Pierce, C. P., & Sayer, A. G. (2007). Shift Work, Role Overload, and the Transition to Parenthood. Journal of Marriage and Family, 69(1), 123–138. http://doi.org/10.1111/j.1741-3737.2006.00349.x
Rodziewicz TL, Hipskind JE. Medical Error Prevention. 2020 May 5. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan–. PMID: 29763131. https://pubmed.ncbi.nlm.nih.gov/29763131/
Tanaka, K., Takahashi, M., Hiro, H., Kakinuma, M., Tanaka, M., Kamata, N., & Miyaoka, H. (2010). Differences in Medical Error Risk among Nurses Working Two- and Three-shift Systems at Teaching Hospitals: A Six-month Prospective Study. Industrial Health, 48(3), 357–364. http://doi.org/10.2486/indhealth.48.357
Admi H, Tzischinsky O, Epstein R, Herer P, Lavie P. Shift work in nursing: is it really a risk factor for nurses’ health and patients’ safety?. Nurs Econ. 2008;26(4):250-257. https://pubmed.ncbi.nlm.nih.gov/18777974/
Clendon, J., & Gibbons, V. (2015). 12h shifts and rates of error among nurses: A systematic review. International Journal of Nursing Studies, 52(7), 1231–1242. http://doi.org/10.1016/j.ijnurstu.2015.03.011
Hammadah, M., Kindya, B. R., Allard‐Ratick, M. P., Jazbeh, S., Eapen, D., Tang, W. W., & Sperling, L. (2017). Navigating air travel and cardiovascular concerns: Is the sky the limit?, Clinical Cardiology, 40 (9), 660–666. http://doi.org/10.1002/clc.22741
Lieber, B. A., Han, J., Appelboom, G., Taylor, B. E., Han, B., Agarwal, N., & Connolly, E. S. (2016). Association of Steroid Use with Deep Venous Thrombosis and Pulmonary Embolism in Neurosurgical Patients: A National Database Analysis. World Neurosurgery, 89, 126–132. http://doi.org/10.1016/j.wneu.2016.01.033
El-Menyar, A., Asim, M., & Al-Thani, H. (2017). Obesity Paradox in Patients With Deep Venous Thrombosis. Clinical and Applied Thrombosis/Hemostasis, 24(6), 986–992. http://doi.org/10.1177/1076029617727858
Klovaite, J., Benn, M., & Nordestgaard, B. G. (2014). Obesity as a causal risk factor for deep venous thrombosis: a Mendelian randomization study. Journal of Internal Medicine, 277(5), 573–584. http://doi.org/10.1111/joim.12299
Davies, H. O., Popplewell, M., Singhal, R., Smith, N., & Bradbury, A. W. (2016). Obesity and lower limb venous disease – The epidemic of phlebesity. Phlebology: The Journal of Venous Disease, 32(4), 227–233. http://doi.org/10.1177/0268355516649333
Liljeqvist, S., Helldén, A., Bergman, U., & Söderberg, M. (2008). Pulmonary embolism associated with the use of anabolic steroids. European Journal of Internal Medicine, 19(3), 214–215. http://doi.org/10.1016/j.ejim.2007.03.016
Linton MF, Yancey PG, Davies SS, Jerome WG (Jay), Linton EF, Vickers KC. The Role of Lipids and Lipoproteins in Atherosclerosis. In: De Groot LJ, Chrousos G, Dungan K, et al., eds. Endotext. South Dartmouth (MA): MDText.com, Inc.; 2000. http://www.ncbi.nlm.nih.gov/books/NBK343489/.
Rescheduling of meals may ease the effects of jet lag. (2017). Nursing Standard, 31(48), 16–16. http://doi.org/10.7748/ns.31.48.16.s17
Ruscitto, C., & Ogden, J. (2016). The impact of an implementation intention to improve mealtimes and reduce jet lag in long-haul cabin crew. Psychology & Health, 32(1), 61–77. http://doi.org/10.1080/08870446.2016.1240174
Reid, K. J., & Abbott, S. M. (2015). Jet Lag and Shift Work Disorder. Sleep Medicine Clinics, 10(4), 523–535. http://doi.org/10.1016/j.jsmc.2015.08.006
Srinivasan, V., Spence, D. W., Pandi-Perumal, S. R., Trakht, I., & Cardinali, D. P. (2008). Jet lag: Therapeutic use of melatonin and possible application of melatonin analogs. Travel Medicine and Infectious Disease, 6(1-2), 17–28. http://doi.org/10.1016/j.tmaid.2007.12.002
Edwards, B. J., Atkinson, G., Waterhouse, J., Reilly, T., Godfrey, R., & Budgett, R. (2000). Use of melatonin in recovery from jet-lag following an eastward flight across 10 time-zones. Ergonomics, 43(10), 1501–1513. http://doi.org/10.1080/001401300750003934
Zee, P. C., & Goldstein, C. A. (2010). Treatment of Shift Work Disorder and Jet Lag. Current Treatment Options in Neurology, 12(5), 396–411. http://doi.org/10.1007/s11940-010-0090-9
https://www.nhlbi.nih.gov/health-topics/circadian-rhythm-disorders
Borodkin, K., & Dagan, Y. (2013). Diagnostic Algorithm for Circadian Rhythm Sleep Disorders. Encyclopedia of Sleep, 66–73. http://doi.org/10.1016/b978-0-12-378610-4.00284-9
Lockley, S. (2013). Special Considerations and Future Directions in Circadian Rhythm Sleep Disorders Diagnosis. Encyclopedia of Sleep, 138–149. http://doi.org/10.1016/b978-0-12-378610-4.00299-0
Crowley, S., & Youngstedt, S. (2013). Pathophysiology, Associations, and Consequences of Circadian Rhythm Sleep Disorder. Encyclopedia of Sleep, 16–21. http://doi.org/10.1016/b978-0-12-378610-4.00266-7
Franken, P., & Dijk, D.-J. (2009). Circadian clock genes and sleep homeostasis. European Journal of Neuroscience, 29(9), 1820–1829. http://doi.org/10.1111/j.1460-9568.2009.06723.x
Burgess, H. J., & Emens, J. S. (2016). Circadian-Based Therapies for Circadian Rhythm Sleep-Wake Disorders. Current Sleep Medicine Reports, 2(3), 158–165. http://doi.org/10.1007/s40675-016-0052-1
Jones, C. R., Huang, A. L., Ptáček, L. J., & Fu, Y.-H. (2013). Genetic basis of human circadian rhythm disorders. Experimental Neurology, 243, 28–33. http://doi.org/10.1016/j.expneurol.2012.07.012
Toh KL. Basic science review on circadian rhythm biology and circadian sleep disorders. Ann Acad Med Singap. 2008;37(8):662-668. https://pubmed.ncbi.nlm.nih.gov/18797559/
Farhud D, Aryan Z. Circadian Rhythm, Lifestyle and Health: A Narrative Review. Iran J Public Health. 2018;47(8):1068-1076. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6123576/
Dodson, E. R., & Zee, P. C. (2010). Therapeutics for Circadian Rhythm Sleep Disorders. Sleep Medicine Clinics, 5(4), 701–715. http://doi.org/10.1016/j.jsmc.2010.08.001
Zhu, L., & Zee, P. C. (2012). Circadian Rhythm Sleep Disorders. Neurologic Clinics, 30(4), 1167–1191. http://doi.org/10.1016/j.ncl.2012.08.011
Kim MJ, Lee JH, Duffy JF. Circadian Rhythm Sleep Disorders. J Clin Outcomes Manag. 2013;20(11):513-528. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4212693/
Zhong, G., Naismith, S. L., Rogers, N. L., & Lewis, S. J. G. (2011). Sleep-wake disturbances in common neurodegenerative diseases: A closer look at selected aspects of the neural circuitry. Journal of the Neurological Sciences, 307(1-2), 9–14. http://doi.org/10.1016/j.jns.2011.04.020
Dijk, D.-J., Boulos, Z., Eastman, C. I., Lewy, A. J., Campbell, S. S., & Terman, M. (1995). Light Treatment for Sleep Disorders: Consensus Report. Journal of Biological Rhythms, 10(2), 113–125. http://doi.org/10.1177/074873049501000204
Barion, A., & Zee, P. C. (2007). A clinical approach to circadian rhythm sleep disorders. Sleep Medicine, 8(6), 566–577. http://doi.org/10.1016/j.sleep.2006.11.017
Horne JA, Ostberg O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int J Chronobiol. 1976;4(2):97-110. https://pubmed.ncbi.nlm.nih.gov/1027738/
Adan, A., & Almirall, H. (1991). Horne & Östberg morningness-eveningness questionnaire: A reduced scale. Personality and Individual Differences, 12(3), 241–253. http://doi.org/10.1016/0191-8869(91)90110-w
Urbán, R., Magyaródi, T., & Rigó, A. (2011). Morningness-Eveningness, Chronotypes and Health-Impairing Behaviors in Adolescents. Chronobiology International, 28(3), 238–247. http://doi.org/10.3109/07420528.2010.549599
https://www.thewep.org/documentations/mctq
Buysse, D. J., Reynolds, C. F., Monk, T. H., Berman, S. R., & Kupfer, D. J. (1989). The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Research, 28(2), 193–213. http://doi.org/10.1016/0165-1781(89)90047-4
Bastien, C. (2001). Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Medicine, 2(4), 297–307. http://doi.org/10.1016/s1389-9457(00)00065-4
Yang, M., Morin, C. M., Schaefer, K., & Wallenstein, G. V. (2009). Interpreting score differences in the Insomnia Severity Index: using health-related outcomes to define the minimally important difference. Current Medical Research and Opinion, 25(10), 2487–2494. http://doi.org/10.1185/03007990903167415
Morin, C. M., Belleville, G., Bélanger, L., & Ivers, H. (2011). The Insomnia Severity Index: Psychometric Indicators to Detect Insomnia Cases and Evaluate Treatment Response. Sleep, 34(5), 601–608. http://doi.org/10.1093/sleep/34.5.601
Castriotta RJ, Wilde MC, Lai JM, Atanasov S, Masel BE, Kuna ST. Prevalence and consequences of sleep disorders in traumatic brain injury. J Clin Sleep Med. 2007;3(4):349-356. https://pubmed.ncbi.nlm.nih.gov/17694722/
Chokroverty S. Overview of sleep & sleep disorders. Indian J Med Res. 2010;131:126-140. https://pubmed.ncbi.nlm.nih.gov/20308738/
Pavlova, M. K., & Latreille, V. (2019). Sleep Disorders. The American Journal of Medicine, 132(3), 292–299. http://doi.org/10.1016/j.amjmed.2018.09.021
Olejniczak, P. W., & Fisch, B. J. (2003). Sleep disorders. Medical Clinics of North America, 87(4), 803–833. http://doi.org/10.1016/s0025-7125(03)00006-3
https://www.nhlbi.nih.gov/health-topics/sleep-apnea
https://clevemed.com/what-is-sleep-apnea/patient-sleep-apnea-screener/
Spicuzza, L., Caruso, D., & Maria, G. D. (2015). Obstructive sleep apnoea syndrome and its management. Therapeutic Advances in Chronic Disease, 6(5), 273–285. http://doi.org/10.1177/2040622315590318
Bixler, E. O., Vgontzas, A. N., Lin, H.-M., Liao, D., Calhoun, S., Fedok, F., … Graff, G. (2008). Blood Pressure Associated With Sleep-Disordered Breathing in a Population Sample of Children. Hypertension, 52(5), 841–846. http://doi.org/10.1161/hypertensionaha.108.116756
Campos, A. I., García-Marín, L. M., Byrne, E. M., Martin, N. G., Cuéllar-Partida, G., & Rentería, M. E. (2020). Insights into the aetiology of snoring from observational and genetic investigations in the UK Biobank. Nature Communications, 11(1). http://doi.org/10.1038/s41467-020-14625-1
Morgenthaler, T. I., Kagramanov, V., Hanak, V., & Decker, P. A. (2006). Complex Sleep Apnea Syndrome: Is It a Unique Clinical Syndrome? Sleep, 29(9), 1203–1209. http://doi.org/10.1093/sleep/29.9.1203
El-Ad, B., & Lavie, P. (2005). Effect of sleep apnea on cognition and mood. International Review of Psychiatry, 17(4), 277–282. http://doi.org/10.1080/09540260500104508
Morgenstern, M., Wang, J., Beatty, N., Batemarco, T., Sica, A. L., & Greenberg, H. (2014). Obstructive Sleep Apnea. Endocrinology and Metabolism Clinics of North America, 43(1), 187–204. http://doi.org/10.1016/j.ecl.2013.09.002
Sleep–Related Breathing Disorders in Adults: Recommendations for Syndrome Definition and Measurement Techniques in Clinical Research. (1999). Sleep, 22(5), 667–689. http://doi.org/10.1093/sleep/22.5.667
Ruehland, W. R., Rochford, P. D., O’Donoghue, F. J., Pierce, R. J., Singh, P., & Thornton, A. T. (2009). The New AASM Criteria for Scoring Hypopneas: Impact on the Apnea Hypopnea Index. Sleep, 32(2), 150–157. http://doi.org/10.1093/sleep/32.2.150
Selim, B. J., Koo, B. B., Qin, L., Jeon, S., Won, C., Redeker, N. S., … Yaggi, H. K. (2016). The Association between Nocturnal Cardiac Arrhythmias and Sleep-Disordered Breathing: The DREAM Study. Journal of Clinical Sleep Medicine, 12(06), 829–837. http://doi.org/10.5664/jcsm.5880
Ahmed, M. H. (2010). Obstructive sleep apnea syndrome and fatty liver: Association or causal link? World Journal of Gastroenterology, 16(34), 4243. http://doi.org/10.3748/wjg.v16.i34.4243
Singh, H., Pollock, R., Uhanova, J., Kryger, M., Hawkins, K., & Minuk, G. Y. (2005). Symptoms of Obstructive Sleep Apnea in Patients with Nonalcoholic Fatty Liver Disease. Digestive Diseases and Sciences, 50(12), 2338–2343. http://doi.org/10.1007/s10620-005-3058-y
Lawati, N. M. A., Patel, S. R., & Ayas, N. T. (2009). Epidemiology, Risk Factors, and Consequences of Obstructive Sleep Apnea and Short Sleep Duration. Progress in Cardiovascular Diseases, 51(4), 285–293. http://doi.org/10.1016/j.pcad.2008.08.001
Young, T. (2004). Risk Factors for Obstructive Sleep Apnea in Adults. Jama, 291(16), 2013. http://doi.org/10.1001/jama.291.16.2013
Yaggi, H. K., Concato, J., Kernan, W. N., Lichtman, J. H., Brass, L. M., & Mohsenin, V. (2005). Obstructive Sleep Apnea as a Risk Factor for Stroke and Death. New England Journal of Medicine, 353(19), 2034–2041. http://doi.org/10.1056/nejmoa043104
Redline, S., Budhiraja, R., Kapur, V., Marcus, C. L., Mateika, J. H., Mehra, R., … Quan, A. S. F. (2007). The Scoring of Respiratory Events in Sleep: Reliability and Validity. Journal of Clinical Sleep Medicine, 03(02), 169–200. http://doi.org/10.5664/jcsm.26818
Basheer B, Hegde KS, Bhat SS, Umar D, Baroudi K. Influence of mouth breathing on the dentofacial growth of children: a cephalometric study. J Int Oral Health. 2014;6(6):50-55. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4295456/
Ruhle, K. H., & Nilius, G. (2008). Mouth Breathing in Obstructive Sleep Apnea prior to and during Nasal Continuous Positive Airway Pressure. Respiration, 76(1), 40–45. http://doi.org/10.1159/000111806
Izu, S. C., Itamoto, C. H., Pradella-Hallinan, M., Pizarro, G. U., Tufik, S., Pignatari, S., & Fujita, R. R. (2010). Obstructive sleep apnea syndrome (OSAS) in mouth breathing children. Brazilian Journal of Otorhinolaryngology, 76(5), 552–556. http://doi.org/10.1590/s1808-86942010000500003
Lee, S. H., Choi, J. H., Shin, C., Lee, H. M., Kwon, S. Y., & Lee, S. H. (2007). How Does Open-Mouth Breathing Influence Upper Airway Anatomy? The Laryngoscope, 117(6), 1102–1106. http://doi.org/10.1097/mlg.0b013e318042aef7
Tuomilehto, H. P. I., Seppä, J. M., Partinen, M. M., Peltonen, M., Gylling, H., Tuomilehto, J. O. I., … Uusitupa, M. (2009). Lifestyle Intervention with Weight Reduction. American Journal of Respiratory and Critical Care Medicine, 179(4), 320–327. http://doi.org/10.1164/rccm.200805-669oc
https://www.cdc.gov/nchs/fastats/obesity-overweight.htm
Neill, A. M., Angus, S. M., Sajkov, D., & Mcevoy, R. D. (1997). Effects of sleep posture on upper airway stability in patients with obstructive sleep apnea. American Journal of Respiratory and Critical Care Medicine, 155(1), 199–204. http://doi.org/10.1164/ajrccm.155.1.9001312
Loord, H., & Hultcrantz, E. (2007). Positioner–a method for preventing sleep apnea. Acta Oto-Laryngologica, 127(8), 861–868. http://doi.org/10.1080/00016480601089390
Szollosi, I., Roebuck, T., Thompson, B., & Naughton, M. T. (2006). Lateral Sleeping Position Reduces Severity of Central Sleep Apnea / Cheyne-Stokes Respiration. Sleep, 29(8), 1045–1051. http://doi.org/10.1093/sleep/29.8.1045
Silverberg DS, Iaina A, Oksenberg A. Treating obstructive sleep apnea improves essential hypertension and quality of life. Am Fam Physician. 2002;65(2):229-236. https://pubmed.ncbi.nlm.nih.gov/11820487/
Aurora, R. N., Chowdhuri, S., Ramar, K., Bista, S. R., Casey, K. R., Lamm, C. I., … Tracy, S. L. (2012). The Treatment of Central Sleep Apnea Syndromes in Adults: Practice Parameters with an Evidence-Based Literature Review and Meta-Analyses. Sleep, 35(1), 17–40. http://doi.org/10.5665/sleep.1580
Hsu, A. A. L., & Lo, C. (2003). Continuous positive airway pressure therapy in sleep apnoea. Respirology, 8(4), 447–454. http://doi.org/10.1046/j.1440-1843.2003.00494.x
Patel, S. R., White, D. P., Malhotra, A., Stanchina, M. L., & Ayas, N. T. (2003). Continuous Positive Airway Pressure Therapy for Treating gess in a Diverse Population With Obstructive Sleep Apnea. Archives of Internal Medicine, 163(5), 565. http://doi.org/10.1001/archinte.163.5.565
Sundaram, S., Lim, J., & Lasserson, T. J. (2005). Surgery for obstructive sleep apnoea in adults. Cochrane Database of Systematic Reviews. http://doi.org/10.1002/14651858.cd001004.pub2
Chen, H., & Lowe, A. A. (2012). Updates in oral appliance therapy for snoring and obstructive sleep apnea. Sleep and Breathing, 17(2), 473–486. http://doi.org/10.1007/s11325-012-0712-4
Gaisl, T., Haile, S. R., Thiel, S., Osswald, M., & Kohler, M. (2019). Efficacy of pharmacotherapy for OSA in adults: A systematic review and network meta-analysis. Sleep Medicine Reviews, 46, 74–86. http://doi.org/10.1016/j.smrv.2019.04.009
Ohayon, M., Wickwire, E. M., Hirshkowitz, M., Albert, S. M., Avidan, A., Daly, F. J., … Vitiello, M. V. (2017). National Sleep Foundations sleep quality recommendations: first report. Sleep Health, 3(1), 6–19. http://doi.org/10.1016/j.sleh.2016.11.006
Youngstedt, S. D., Goff, E. E., Reynolds, A. M., Kripke, D. F., Irwin, M. R., Bootzin, R. R., … Jean-Louis, G. (2016). Has adult sleep duration declined over the last 50 years? Sleep Medicine Reviews, 28, 69–85. http://doi.org/10.1016/j.smrv.2015.08.004
Chaput, J.-P., Mcneil, J., Després, J.-P., Bouchard, C., & Tremblay, A. (2013). Seven to Eight Hours of Sleep a Night Is Associated with a Lower Prevalence of the Metabolic Syndrome and Reduced Overall Cardiometabolic Risk in Adults. PLoS ONE, 8(9). http://doi.org/10.1371/journal.pone.0072832
Wild, C. J., Nichols, E. S., Battista, M. E., Stojanoski, B., & Owen, A. M. (2018). Dissociable effects of self-reported daily sleep duration on high-level cognitive abilities. Sleep, 41(12). http://doi.org/10.1093/sleep/zsy182
Hirshkowitz, M., Whiton, K., Albert, S. M., Alessi, C., Bruni, O., Doncarlos, L., … Hillard, P. J. A. (2015). National Sleep Foundation’s sleep time duration recommendations: methodology and results summary. Sleep Health, 1(1), 40–43. http://doi.org/10.1016/j.sleh.2014.12.010
Cappuccio, F. P., Delia, L., Strazzullo, P., & Miller, M. A. (2010). Sleep Duration and All-Cause Mortality: A Systematic Review and Meta-Analysis of Prospective Studies. Sleep, 33(5), 585–592. http://doi.org/10.1093/sleep/33.5.585
Gottlieb, D. J., Punjabi, N. M., Newman, A. B., Resnick, H. E., Redline, S., Baldwin, C. M., & Nieto, F. J. (2005). Association of Sleep Time With Diabetes Mellitus and Impaired Glucose Tolerance. Archives of Internal Medicine, 165(8), 863. http://doi.org/10.1001/archinte.165.8.863
Short, M. A., Agostini, A., Lushington, K., & Dorrian, J. (2015). A systematic review of the sleep, sleepiness, and performance implications of limited wake shift work schedules. Scandinavian Journal of Work, Environment & Health, 41(5), 425–440. http://doi.org/10.5271/sjweh.3509
Cappuccio, F. P., Taggart, F. M., Kandala, N.-B., Currie, A., Peile, E., Stranges, S., & Miller, M. A. (2008). Meta-Analysis of Short Sleep Duration and Obesity in Children and Adults. Sleep, 31(5), 619–626. http://doi.org/10.1093/sleep/31.5.619
Mong, J. A., & Cusmano, D. M. (2016). Sex differences in sleep: impact of biological sex and sex steroids. Philosophical Transactions of the Royal Society B: Biological Sciences, 371(1688), 20150110. http://doi.org/10.1098/rstb.2015.0110
Krishnan, V., & Collop, N. A. (2006). Gender differences in sleep disorders. Current Opinion in Pulmonary Medicine, 12(6), 383–389. http://doi.org/10.1097/01.mcp.0000245705.69440.6a
Mehta, N., Shafi, F., & Bhat, A. (2015). Unique Aspects of Sleep in Women. Missouri medicine, 112(6), 430–434. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6168103/
Moline, M. L., Broch, L., & Zak, R. (2004). Sleep in women across the life cycle from adulthood through menopause. Medical Clinics of North America, 88(3), 705–736. http://doi.org/10.1016/j.mcna.2004.01.009
He, Y., Jones, C. R., Fujiki, N., Xu, Y., Guo, B., Holder, J. L., … Fu, Y.-H. (2009). The Transcriptional Repressor DEC2 Regulates Sleep Length in Mammals. Science, 325(5942), 866–870. http://doi.org/10.1126/science.1174443
Theorell-Haglöw, J., Berglund, L., Berne, C., & Lindberg, E. (2014). Both habitual short sleepers and long sleepers are at greater risk of obesity: a population-based 10-year follow-up in women. Sleep Medicine, 15(10), 1204–1211. http://doi.org/10.1016/j.sleep.2014.02.014
Mezick, E. J., Wing, R. R., & Mccaffery, J. M. (2014). Associations of self-reported and actigraphy-assessed sleep characteristics with body mass index and waist circumference in adults: moderation by gender. Sleep Medicine, 15(1), 64–70. http://doi.org/10.1016/j.sleep.2013.08.784
Kim, S. J. (2011). Relationship Between Weekend Catch-up Sleep and Poor Performance on Attention Tasks in Korean Adolescents. Archives of Pediatrics & Adolescent Medicine, 165(9), 806. http://doi.org/10.1001/archpediatrics.2011.128
Kim, C.-W., Choi, M.-K., Im, H.-J., Kim, O.-H., Lee, H.-J., Song, J., … Park, K.-H. (2012). Weekend catch-up sleep is associated with decreased risk of being overweight among fifth-grade students with short sleep duration. Journal of Sleep Research, 21(5), 546–551. http://doi.org/10.1111/j.1365-2869.2012.01013.x
Sun, W., Ling, J., Zhu, X., Lee, T. M.-C., & Li, S. X. (2019). Associations of weekday-to-weekend sleep differences with academic performance and health-related outcomes in school-age children and youths. Sleep Medicine Reviews, 46, 27–53. http://doi.org/10.1016/j.smrv.2019.04.003
Kang, S.-G., Lee, Y. J., Kim, S. J., Lim, W., Lee, H.-J., Park, Y.-M., … Hong, J. P. (2014). Weekend catch-up sleep is independently associated with suicide attempts and self-injury in Korean adolescents. Comprehensive Psychiatry, 55(2), 319–325. http://doi.org/10.1016/j.comppsych.2013.08.023
Zhao, M., Tuo, H., Wang, S., & Zhao, L. (2020). The Effects of Dietary Nutrition on Sleep and Sleep Disorders. Mediators of Inflammation, 2020, 1–7. http://doi.org/10.1155/2020/3142874
Doherty, Madigan, Warrington, & Ellis. (2019). Sleep and Nutrition Interactions: Implications for Athletes. Nutrients, 11(4), 822. http://doi.org/10.3390/nu11040822
Sutanto CN, Wang MX, Tan D, Kim JE. Association of Sleep Quality and Macronutrient Distribution: A Systematic Review and Meta-Regression. Nutrients. 2020 Jan 2;12(1):126. doi: 10.3390/nu12010126. PMID: 31906452; PMCID: PMC7019667. https://pubmed.ncbi.nlm.nih.gov/31906452/
Peuhkuri, K., Sihvola, N., & Korpela, R. (2012). Diet promotes sleep duration and quality. Nutrition Research, 32(5), 309–319. http://doi.org/10.1016/j.nutres.2012.03.009
Afaghi, A., Oconnor, H., & Chow, C. M. (2007). High-glycemic-index carbohydrate meals shorten sleep onset. The American Journal of Clinical Nutrition, 85(2), 426–430. http://doi.org/10.1093/ajcn/85.2.426
Golem, D. L., Martin-Biggers, J. T., Koenings, M. M., Davis, K. F., & Byrd-Bredbenner, C. (2014). An Integrative Review of Sleep for Nutrition Professionals. Advances in Nutrition, 5(6), 742–759. http://doi.org/10.3945/an.114.006809
Grandner, M. A., Jackson, N., Gerstner, J. R., & Knutson, K. L. (2013). Sleep symptoms associated with intake of specific dietary nutrients. Journal of Sleep Research, 23(1), 22–34. http://doi.org/10.1111/jsr.12084
Porter, J., & Horne, J. (1981). Bed-time food supplements and sleep: Effects of different carbohydrate levels. Electroencephalography and Clinical Neurophysiology, 51(4), 426–433. http://doi.org/10.1016/0013-4694(81)90106-1
Halson, S. L. (2014). Sleep in Elite Athletes and Nutritional Interventions to Enhance Sleep. Sports Medicine, 44(S1), 13–23. http://doi.org/10.1007/s40279-014-0147-0
Rondanelli, M., Opizzi, A., Monteferrario, F., Antoniello, N., Manni, R., & Klersy, C. (2011). The Effect of Melatonin, Magnesium, and Zinc on Primary Insomnia in Long-Term Care Facility Residents in Italy: A Double-Blind, Placebo-Controlled Clinical Trial. Journal of the American Geriatrics Society, 59(1), 82–90. http://doi.org/10.1111/j.1532-5415.2010.03232.x
Tahara, Y., & Shibata, S. (2014). Chrono-biology, Chrono-pharmacology, and Chrono-nutrition. Journal of Pharmacological Sciences, 124(3), 320–335. http://doi.org/10.1254/jphs.13r06cr
Landolt, H.-P., Werth, E., Borbély, A. A., & Dijk, D.-J. (1995). Caffeine intake (200 mg) in the morning affects human sleep and EEG power spectra at night. Brain Research, 675(1-2), 67–74. http://doi.org/10.1016/0006-8993(95)00040-w
Gottesmann, C. (2002). GABA mechanisms and sleep. Neuroscience, 111(2), 231–239. http://doi.org/10.1016/s0306-4522(02)00034-9
Campbell, S. S., Dawson, D., & Anderson, M. W. (1993). Alleviation of Sleep Maintenance Insomnia with Timed Exposure to Bright Light. Journal of the American Geriatrics Society, 41(8), 829–836. http://doi.org/10.1111/j.1532-5415.1993.tb06179.x
Zhao, J., Tian, Y., Nie, J., Xu, J., & Liu, D. (2012). Red Light and the Sleep Quality and Endurance Performance of Chinese Female Basketball Players. Journal of Athletic Training, 47(6), 673–678. http://doi.org/10.4085/1062-6050-47.6.08
Smolensky, M. H., Sackett-Lundeen, L. L., & Portaluppi, F. (2015). Nocturnal light pollution and underexposure to daytime sunlight: Complementary mechanisms of circadian disruption and related diseases. Chronobiology International, 32(8), 1029–1048. http://doi.org/10.3109/07420528.2015.1072002
Düzgün, G., & Akyol, A. D. (2017). Effect of Natural Sunlight on Sleep Problems and Sleep Quality of the Elderly Staying in the Nursing Home. Holistic Nursing Practice, 31(5), 295–302. http://doi.org/10.1097/hnp.0000000000000206
Valham, F., Sahlin, C., Stenlund, H., & Franklin, K. A. (2012). Ambient Temperature and Obstructive Sleep Apnea: Effects on Sleep, Sleep Apnea, and Morning Alertness. Sleep, 35(4), 513–517. http://doi.org/10.5665/sleep.1736
Okamoto-Mizuno, K., Tsuzuki, K., & Mizuno, K. (2004). Effects of mild heat exposure on sleep stages and body temperature in older men. International Journal of Biometeorology, 49(1). http://doi.org/10.1007/s00484-004-0209-3
St-Onge, M.-P., & Shechter, A. (2014). Sleep disturbances, body fat distribution, food intake and/or energy expenditure: pathophysiological aspects. Hormone Molecular Biology and Clinical Investigation, 17(1). http://doi.org/10.1515/hmbci-2013-0066
Chaput, J.-P., Després, J.-P., Bouchard, C., & Tremblay, A. (2008). The Association Between Sleep Duration and Weight Gain in Adults: A 6-Year Prospective Study from the Quebec Family Study. Sleep, 31(4), 517–523. http://doi.org/10.1093/sleep/31.4.517
Dekker, S. A., Noordam, R., Biermasz, N. R., Roos, A., Lamb, H. J., Rosendaal, F. R., … Mutsert, R. (2018). Habitual Sleep Measures are Associated with Overall Body Fat, and not Specifically with Visceral Fat, in Men and Women. Obesity, 26(10), 1651–1658. http://doi.org/10.1002/oby.22289
Tunnicliffe, J. M., Erdman, K. A., Reimer, R. A., Lun, V., & Shearer, J. (2008). Consumption of dietary caffeine and coffee in physically active populations: physiological interactions. Applied Physiology, Nutrition, and Metabolism, 33(6), 1301–1310. http://doi.org/10.1139/h08-124
Mahoney, C. R., Giles, G. E., Marriott, B. P., Judelson, D. A., Glickman, E. L., Geiselman, P. J., & Lieberman, H. R. (2019). Intake of caffeine from all sources and reasons for use by college students. Clinical Nutrition, 38(2), 668–675. http://doi.org/10.1016/j.clnu.2018.04.004
Binks, H., Vincent, G. E., Gupta, C., Irwin, C., & Khalesi, S. (2020). Effects of Diet on Sleep: A Narrative Review. Nutrients, 12(4), 936. http://doi.org/10.3390/nu12040936
Rao, T. P., Ozeki, M., & Juneja, L. R. (2015). In Search of a Safe Natural Sleep Aid. Journal of the American College of Nutrition, 34(5), 436–447. http://doi.org/10.1080/07315724.2014.926153
Abbasi B, Kimiagar M, Sadeghniiat K, Shirazi MM, Hedayati M, Rashidkhani B. The effect of magnesium supplementation on primary insomnia in elderly: A double-blind placebo-controlled clinical trial. J Res Med Sci. 2012 Dec;17(12):1161-9. https://pubmed.ncbi.nlm.nih.gov/23853635/
Aspy, D. J., Madden, N. A., & Delfabbro, P. (2018). Effects of Vitamin B6 (Pyridoxine) and a B Complex Preparation on Dreaming and Sleep. Perceptual and Motor Skills, 003151251877032. http://doi.org/10.1177/0031512518770326
Parazzini F. Resveratrol, tryptophanum, glycine and vitamin E: a nutraceutical approach to sleep disturbance and irritability in peri- and post-menopause. Minerva Ginecol. 2015;67(1):1-5. https://pubmed.ncbi.nlm.nih.gov/25660429/
Siegel JM. The neurotransmitters of sleep. J Clin Psychiatry. 2004;65 Suppl 16:4-7. https://pubmed.ncbi.nlm.nih.gov/15575797/
Djeridane, Y., Touitou, Y. Chronic diazepam administration differentially affects melatonin synthesis in rat pineal and Harderian glands. Psychopharmacology154, 403–407 (2001). https://doi.org/10.1007/s002130000631
Betti L, Palego L, Demontis GC, Miraglia F, Giannaccini G. Hydroxyindole-O-methyltransferase (HIOMT) activity in the retina of melatonin-proficient mice. Heliyon. 2019;5(9):e02417. Published 2019 Sep 14. doi:10.1016/j.heliyon.2019.e02417
Haduch, A., Bromek, E., Wójcikowski, J., Gołembiowska, K., Daniel, W.. Melatonin Supports Serotonin Formation by Brain CYP2D. Drug Metabolism and DispositionMarch 1, 2016, 44 (3) 445-452; DOI: https://doi.org/10.1124/dmd.115.067413
Morton, D. J. (1987). Mechanism of Inhibition of Bovine Pineal Gland Hydroxyindole-O-Methyltransferase (EC 2.1.1.4) by Divalent Cations. Journal of Pineal Research, 4(3), 295–303. http://doi.org/10.1111/j.1600-079x.1987.tb00867.x
Markova-Car, E. P., Jurišić, D., Ilić, N., & Pavelić, S. K. (2014). Running for time: circadian rhythms and melanoma. Tumor Biology, 35(9), 8359–8368. http://doi.org/10.1007/s13277-014-1904-2
Slominski AT, Zmijewski MA, Skobowiat C, Zbytek B, Slominski RM, Steketee JD. Sensing the environment: regulation of local and global homeostasis by the skin’s neuroendocrine system. Adv Anat Embryol Cell Biol. 2012;212:v-115. doi:10.1007/978-3-642-19683-6_1 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3422784/
Chalupsky, K., Kračun, D., Kanchev, I., Bertram, K., & Görlach, A. (2015). Folic Acid Promotes Recycling of Tetrahydrobiopterin and Protects Against Hypoxia-Induced Pulmonary Hypertension by Recoupling Endothelial Nitric Oxide Synthase. Antioxidants & Redox Signaling, 23(14), 1076–1091. http://doi.org/10.1089/ars.2015.6329
Dianzani, I., Sanctis, L. D., Smooker, P. M., Gough, T. J., Alliaudi, C., Brusco, A., … Cotton, R. G. H. (1998). Dihydropteridine reductase deficiency: Physical structure of the QDPR gene, identification of two new mutations and genotype–phenotype correlations. Human Mutation, 12(4), 267–273. http://doi.org/10.1002/(sici)1098-1004(1998)12:4<267::aid-humu8>3.0.co;2-c
Nichol, C. A., Lee, C. L., Edelstein, M. P., Chao, J. Y., & Duch, D. S. (1983). Biosynthesis of tetrahydrobiopterin by de novo and salvage pathways in adrenal medulla extracts, mammalian cell cultures, and rat brain in vivo. Proceedings of the National Academy of Sciences, 80(6), 1546–1550. http://doi.org/10.1073/pnas.80.6.1546
Titus, F., Dávalos, A., Alom, J., & Codina, A. (1986). 5-Hydroxytryptophan versus Methysergide in the Prophylaxis of Migraine. European Neurology, 25(5), 327–329. http://doi.org/10.1159/000116030
Birdsall TC. 5-Hydroxytryptophan: a clinically-effective serotonin precursor. Altern Med Rev. 1998;3(4):271-280. https://pubmed.ncbi.nlm.nih.gov/9727088/
Volpi-Abadie J, Kaye AM, Kaye AD. Serotonin syndrome. Ochsner J. 2013;13(4):533-540. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3865832/
Valadas, J. S., Esposito, G., Vandekerkhove, D., Miskiewicz, K., Deaulmerie, L., Raitano, S., … Verstreken, P. (2018). ER Lipid Defects in Neuropeptidergic Neurons Impair Sleep Patterns in Parkinson’s Disease. Neuron, 98(6). http://doi.org/10.1016/j.neuron.2018.05.022
Chung SY, Moriyama T, Uezu E, et al. Administration of phosphatidylcholine increases brain acetylcholine concentration and improves memory in mice with dementia. J Nutr. 1995;125(6):1484-1489. doi:10.1093/jn/125.6.1484 https://pubmed.ncbi.nlm.nih.gov/7782901/
Montgomery, P., Burton, J. R., Sewell, R. P., Spreckelsen, T. F., & Richardson, A. J. (2014). Fatty acids and sleep in UK children: subjective and pilot objective sleep results from the DOLAB study – a randomized controlled trial. Journal of Sleep Research, 23(4), 364–388. http://doi.org/10.1111/jsr.12135
Alzoubi, K. H., Mayyas, F., & Zamzam, H. I. A. (2019). Omega-3 fatty acids protects against chronic sleep-deprivation induced memory impairment. Life Sciences, 227, 1–7. http://doi.org/10.1016/j.lfs.2019.04.028
Nasehi, M., Mosavi-Nezhad, S.-M., Khakpai, F., & Zarrindast, M.-R. (2018). The role of omega-3 on modulation of cognitive deficiency induced by REM sleep deprivation in rats. Behavioural Brain Research, 351, 152–160. http://doi.org/10.1016/j.bbr.2018.06.002
Jahangard, L., Sadeghi, A., Ahmadpanah, M., Holsboer-Trachsler, E., Bahmani, D. S., Haghighi, M., & Brand, S. (2018). Influence of adjuvant omega-3-polyunsaturated fatty acids on depression, sleep, and emotion regulation among outpatients with major depressive disorders – Results from a double-blind, randomized and placebo-controlled clinical trial. Journal of Psychiatric Research, 107, 48–56. http://doi.org/10.1016/j.jpsychires.2018.09.016
Scorza, F. A., Cavalheiro, E. A., Scorza, C. A., Galduróz, J. C. F., Tufik, S., & Andersen, M. L. (2013). Sleep Apnea and Inflammation – Getting a Good Night’s Sleep with Omega-3 Supplementation. Frontiers in Neurology, 4. http://doi.org/10.3389/fneur.2013.00193
Hansen, A. L., Dahl, L., Olson, G., Thornton, D., Graff, I. E., Frøyland, L., … Pallesen, S. (2014). Fish Consumption, Sleep, Daily Functioning, and Heart Rate Variability. Journal of Clinical Sleep Medicine, 10(05), 567–575. http://doi.org/10.5664/jcsm.3714
Xie, L., Kang, H., Xu, Q., Chen, M. J., Liao, Y., Thiyagarajan, M., … Nedergaard, M. (2013). Sleep Drives Metabolite Clearance from the Adult Brain. Science, 342(6156), 373–377. http://doi.org/10.1126/science.1241224
Varshavsky, A. (2012). Augmented generation of protein fragments during wakefulness as the molecular cause of sleep: a hypothesis. Protein Science, 21(11), 1634–1661. http://doi.org/10.1002/pro.2148
Mackiewicz, M., Shockley, K. R., Romer, M. A., Galante, R. J., Zimmerman, J. E., Naidoo, N., … Pack, A. I. (2007). Macromolecule biosynthesis: a key function of sleep. Physiological Genomics, 31(3), 441–457. http://doi.org/10.1152/physiolgenomics.00275.2006
Scharf, M. T., Naidoo, N., Zimmerman, J. E., & Pack, A. I. (2008). The energy hypothesis of sleep revisited. Progress in Neurobiology, 86(3), 264–280. http://doi.org/10.1016/j.pneurobio.2008.08.003
Horne, J. (2009). REM sleep, energy balance and ‘optimal foraging.’ Neuroscience & Biobehavioral Reviews, 33(3), 466–474. http://doi.org/10.1016/j.neubiorev.2008.12.002
Berger, R. J., & Phillips, N. H. (1995). Energy conservation and sleep. Behavioural Brain Research, 69(1-2), 65–73. http://doi.org/10.1016/0166-4328(95)00002-b
Benington, J. H., & Heller, H. C. (1995). Restoration of brain energy metabolism as the function of sleep. Progress in Neurobiology, 45(4), 347–360. http://doi.org/10.1016/0301-0082(94)00057-o
Abel, T., Havekes, R., Saletin, J. M., & Walker, M. P. (2013). Sleep, Plasticity and Memory from Molecules to Whole-Brain Networks. Current Biology, 23(17). http://doi.org/10.1016/j.cub.2013.07.025
Rasch, B., & Born, J. (2013). About Sleeps Role in Memory. Physiological Reviews, 93(2), 681–766. http://doi.org/10.1152/physrev.00032.2012
Cirelli, C., & Tononi, G. (2008). Is Sleep Essential? PLoS Biology, 6(8). http://doi.org/10.1371/journal.pbio.0060216
Siegel, J. M. (2005). Clues to the functions of mammalian sleep. Nature, 437(7063), 1264–1271. http://doi.org/10.1038/nature04285
Campbell, S. S., & Tobler, I. (1984). Animal sleep: A review of sleep duration across phylogeny. Neuroscience & Biobehavioral Reviews, 8(3), 269–300. http://doi.org/10.1016/0149-7634(84)90054-x
Tobler, I. (1995). Is sleep fundamentally different between mammalian species? Behavioural Brain Research, 69(1-2), 35–41. http://doi.org/10.1016/0166-4328(95)00025-o
Tononi, G., & Cirelli, C. (2006). Sleep function and synaptic homeostasis. Sleep Medicine Reviews, 10(1), 49–62. http://doi.org/10.1016/j.smrv.2005.05.002
Dongen, H. P. A. V., Vitellaro, K. M., & Dinges, D. F. (2005). Individual Differences in Adult Human Sleep and Wakefulness: Leitmotif for a Research Agenda. Sleep, 28(4), 479–498. http://doi.org/10.1093/sleep/28.4.479
Vyazovskiy, V. V., & Delogu, A. (2014). NREM and REM Sleep. The Neuroscientist, 20(3), 203–219. http://doi.org/10.1177/1073858413518152
Mignot, E. (2008). Why We Sleep: The Temporal Organization of Recovery. PLoS Biology, 6(4). http://doi.org/10.1371/journal.pbio.0060106
Siegel, J. M. (2009). Sleep viewed as a state of adaptive inactivity. Nature Reviews Neuroscience, 10(10), 747–753. http://doi.org/10.1038/nrn2697
Horne, J. (2000). REM sleep — by default? Neuroscience & Biobehavioral Reviews, 24(8), 777–797. http://doi.org/10.1016/s0149-7634(00)00037-3
Baran, B., Pace-Schott, E. F., Ericson, C., & Spencer, R. M. C. (2012). Processing of Emotional Reactivity and Emotional Memory over Sleep. Journal of Neuroscience, 32(3), 1035–1042. http://doi.org/10.1523/jneurosci.2532-11.2012
Tononi, G., & Cirelli, C. (2014). Sleep and the Price of Plasticity: From Synaptic and Cellular Homeostasis to Memory Consolidation and Integration. Neuron, 81(1), 12–34. http://doi.org/10.1016/j.neuron.2013.12.025
Li, J., Vitiello, M. V., & Gooneratne, N. S. (2018). Sleep in Normal Aging. Sleep Medicine Clinics, 13(1), 1–11. http://doi.org/10.1016/j.jsmc.2017.09.001
Murillo-Rodriguez, E., Arias-Carrion, O., Zavala-Garcia, A., Sarro-Ramirez, A., & Huitron-Resendiz, S. (2012). Basic Sleep Mechanisms: An Integrative Review. Central Nervous System Agents in Medicinal Chemistry, 12(1), 38–54. http://doi.org/10.2174/187152412800229107
Weber, F. D. (2018). Sleep: Eye-Opener Highlights Sleep’s Organization. Current Biology, 28(5). http://doi.org/10.1016/j.cub.2018.01.054
Yetton, B. D., Mcdevitt, E. A., Cellini, N., Shelton, C., & Mednick, S. C. (2018). Quantifying sleep architecture dynamics and individual differences using big data and Bayesian networks. Plos One, 13(4). http://doi.org/10.1371/journal.pone.0194604
Colrain, I. M., Nicholas, C. L., & Baker, F. C. (2014). Alcohol and the sleeping brain. Handbook of Clinical Neurology Alcohol and the Nervous System, 415–431. http://doi.org/10.1016/b978-0-444-62619-6.00024-0
Zisapel, N. (2018). New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. British Journal of Pharmacology, 175(16), 3190–3199. http://doi.org/10.1111/bph.14116
Tosini, G., Baba, K., Hwang, C. K., & Iuvone, P. M. (2012). Melatonin: An underappreciated player in retinal physiology and pathophysiology. Experimental Eye Research, 103, 82–89. http://doi.org/10.1016/j.exer.2012.08.009
Blasiak, J., Reiter, R. J., & Kaarniranta, K. (2016). Melatonin in Retinal Physiology and Pathology: The Case of Age-Related Macular Degeneration. Oxidative Medicine and Cellular Longevity, 2016, 1–12. http://doi.org/10.1155/2016/6819736
Bellingham, J., Chaurasia, S. S., Melyan, Z., Liu, C., Cameron, M. A., Tarttelin, E. E., … Lucas, R. J. (2006). Evolution of Melanopsin Photoreceptors: Discovery and Characterization of a New Melanopsin in Nonmammalian Vertebrates. PLoS Biology, 4(8). http://doi.org/10.1371/journal.pbio.0040254
Zaidi, F. H., Hull, J. T., Peirson, S. N., Wulff, K., Aeschbach, D., Gooley, J. J., … Lockley, S. W. (2007). Short-Wavelength Light Sensitivity of Circadian, Pupillary, and Visual Awareness in Humans Lacking an Outer Retina. Current Biology, 17(24), 2122–2128. http://doi.org/10.1016/j.cub.2007.11.034
Legates, T. A., Altimus, C. M., Wang, H., Lee, H.-K., Yang, S., Zhao, H., … Hattar, S. (2012). Aberrant light directly impairs mood and learning through melanopsin-expressing neurons. Nature, 491(7425), 594–598. http://doi.org/10.1038/nature11673
Sikka, G., Hussmann, G. P., Pandey, D., Cao, S., Hori, D., Park, J. T., … Berkowitz, D. E. (2014). Melanopsin mediates light-dependent relaxation in blood vessels. Proceedings of the National Academy of Sciences, 111(50), 17977–17982. http://doi.org/10.1073/pnas.1420258111
Buhr, E. D., Yoo, S.-H., & Takahashi, J. S. (2010). Temperature as a Universal Resetting Cue for Mammalian Circadian Oscillators. Science, 330(6002), 379–385. http://doi.org/10.1126/science.1195262
Robbins, R., Grandner, M. A., Buxton, O. M., Hale, L., Buysse, D. J., Knutson, K. L., … Jean-Louis, G. (2019). Sleep myths: an expert-led study to identify false beliefs about sleep that impinge upon population sleep health practices. Sleep Health, 5(4), 409–417. http://doi.org/10.1016/j.sleh.2019.02.002
Damiola, F. (2000). Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes & Development, 14(23), 2950–2961. http://doi.org/10.1101/gad.183500
Abel, T., Havekes, R., Saletin, J. M., & Walker, M. P. (2013). Sleep, Plasticity and Memory from Molecules to Whole-Brain Networks. Current Biology, 23(17). http://doi.org/10.1016/j.cub.2013.07.025
Muzet, A., Ehrhart, J., Candas, V., Libert, J. P., & Vogt, J. J. (1983). Rem Sleep and Ambient Temperature in Man. International Journal of Neuroscience, 18(1-2), 117–125. http://doi.org/10.3109/00207458308985885
Saini, C., Morf, J., Stratmann, M., Gos, P., & Schibler, U. (2012). Simulated body temperature rhythms reveal the phase-shifting behavior and plasticity of mammalian circadian oscillators. Genes & Development, 26(6), 567–580. http://doi.org/10.1101/gad.183251.111
Franco P, Szliwowski H, Dramaix M, Kahn A. Influence of ambient temperature on sleep characteristics and autonomic nervous control in healthy infants. Sleep. 2000 May 1;23(3):401-7. https://pubmed.ncbi.nlm.nih.gov/10811384/
Libert JP, Candas V, Muzet A, Ehrhart J. Thermoregulatory adjustments to thermal transients during slow wave sleep and REM sleep in man. J Physiol (Paris). 1982;78(3):251-7 https://pubmed.ncbi.nlm.nih.gov/7166740/
Palca, J. W., Walker, J. M., & Berger, R. J. (1986). Thermoregulation, metabolism, and stages of sleep in cold-exposed men. Journal of Applied Physiology, 61(3), 940–947. http://doi.org/10.1152/jappl.1986.61.3.940
Lack. (2009). Chronotype differences in circadian rhythms of temperature, melatonin, and sleepiness as measured in a modified constant routine protocol. Nature and Science of Sleep, 1. http://doi.org/10.2147/nss.s6234
Samson, D. R., Crittenden, A. N., Mabulla, I. A., Mabulla, A. Z. P., & Nunn, C. L. (2017). Chronotype variation drives night-time sentinel-like behaviour in hunter–gatherers. Proceedings of the Royal Society B: Biological Sciences, 284(1858), 20170967. http://doi.org/10.1098/rspb.2017.0967
Walker, R. J., Kribs, Z. D., Christopher, A. N., Shewach, O. R., & Wieth, M. B. (2014). Age, the Big Five, and time-of-day preference: A mediational model. Personality and Individual Differences, 56, 170–174. http://doi.org/10.1016/j.paid.2013.09.003
Bjorness, T., & Greene, R. (2009). Adenosine and Sleep. Current Neuropharmacology, 7(3), 238–245. http://doi.org/10.2174/157015909789152182
Dworak, M., Diel, P., Voss, S., Hollmann, W., & Strüder, H. (2007). Intense exercise increases adenosine concentrations in rat brain: Implications for a homeostatic sleep drive. Neuroscience, 150(4), 789–795. http://doi.org/10.1016/j.neuroscience.2007.09.062
Rainnie, D., Grunze, H., Mccarley, R., & Greene, R. (1994). Adenosine inhibition of mesopontine cholinergic neurons: implications for EEG arousal. Science, 263(5147), 689–692. http://doi.org/10.1126/science.8303279
Daly, J. W., Shi, D., Nikodijevic, O., & Jacobson, K. A. (1994). The role of adenosine receptors in the central action of caffeine. Pharmacopsychoecologia, 7(2), 201–213. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4373791/

